Inverse opal substrate-loaded mesenchymal stem cells contribute to decreased myocardial remodeling after transplantation into acute myocardial infarction mice
- PMID: 30464457
- PMCID: PMC6220438
- DOI: 10.2147/IJN.S178270
Inverse opal substrate-loaded mesenchymal stem cells contribute to decreased myocardial remodeling after transplantation into acute myocardial infarction mice
Abstract
Background: The two-dimensional incubation method is now the most commonly method for mesenchymal stem cell (MSC) production. however, gene expression and secretion of growth factors are relatively low; thus, the transplanted cells cannot be effectively utilized for potential clinical applications after acute myocardial infarction (AMI).
Objectives: We aimed to investigate whether our newly made substrates of inverse opal with specific surface microstructures for MSC culturing can increase the viability of the cells and can contributes to decreased myocardial remodeling after transplanted to AMI mice.
Methods: The inverse opal structure is fabricated by the convenient bottom-up approach of the self-assembly of colloidal nanoparticles. Mouse-derived MSCs were then cultured on the substrates when expanded at different times to investigate the cell growth status including morphology. Then the inverse opal substrates loaded MSCs were transplanted to AMI mice, cardiomyocyte apoptosis and LV remodeling were further compared. To explore the possible mechanisms of curation, the secretions and viability of MSCs on substrates were determined using mice ELISA kits and JC-1 mitochondrial membrane potential assay kits respectively at normal and hypoxic conditions.
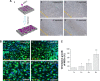
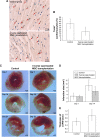
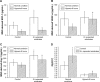
Results: 6 times expanded inverse opals allowed greatly the orderly growth of MSCs as compared to four (34% ± 10.6%) and two (20%±7.2%) times expanded as well as unexpanded (13%±4.1%) (P<0.001). Nearly 90% of MSCs showed orientation angle intervals of less than 30° when at the 6X expanded (89.6%±25%) compared to the percent of cells with 30°-60° (8.7%±2.6%) or ≥60° (1.7%±1.0%) orientation angle (P<0.001). After inverse opal loaded MSCs transplanted to AMI mice, greatly decreased apoptosis of cardiomyocytes (20.45%±8.64% vs.39.63%±11.71%, P<0.001) and infarction area (5.87±2.18 mm2 vs 9.31±3.11 mm2, P<0.001) were identified. In the end, the viability of inverse opal loaded MSCs determined by membrane potential (P<0.001) and the secretion of growth factors including VEGF-α, SDF-1 and Ang-1 (P<0.001) were both confirmed significantly higher than that of the conventional culture in petri dish.
Conclusion: The structure of inverse opal can not only adjust the arrangement of MSCs but also contribute to its orientated growth. Inverse opal loaded MSCs transplantation extremely curbed myocardial remodeling, the underlying mechanisms might be the high viability and extremely higher secretions of growth factors of MSCs as devoted by this method.
Keywords: AMI; MSCs; inverse opal.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures




Similar articles
-
Photoluminescent Mesoporous Silicon Nanoparticles with siCCR2 Improve the Effects of Mesenchymal Stromal Cell Transplantation after Acute Myocardial Infarction.Theranostics. 2015 Jun 25;5(10):1068-82. doi: 10.7150/thno.11517. eCollection 2015. Theranostics. 2015. PMID: 26199646 Free PMC article.
-
Effect of transplanted mesenchymal stem cells from rats of different ages on the improvement of heart function after acute myocardial infarction.Chin Med J (Engl). 2008 Nov 20;121(22):2290-8. Chin Med J (Engl). 2008. PMID: 19080335
-
Mesenchymal stem cells modified with stromal cell-derived factor 1 alpha improve cardiac remodeling via paracrine activation of hepatocyte growth factor in a rat model of myocardial infarction.Mol Cells. 2010 Jan;29(1):9-19. doi: 10.1007/s10059-010-0001-7. Epub 2009 Dec 7. Mol Cells. 2010. PMID: 20016947
-
The Vital Roles of Mesenchymal Stem Cells and the Derived Extracellular Vesicles in Promoting Angiogenesis After Acute Myocardial Infarction.Stem Cells Dev. 2021 Jun 1;30(11):561-577. doi: 10.1089/scd.2021.0006. Epub 2021 Apr 30. Stem Cells Dev. 2021. PMID: 33752473 Review.
-
Research Progress on Cardiac Tissue Construction of Mesenchymal Stem Cells for Myocardial Infarction.Curr Stem Cell Res Ther. 2024;19(7):942-958. doi: 10.2174/1574888X18666230823091017. Curr Stem Cell Res Ther. 2024. PMID: 37612870 Review.
Cited by
-
MSCs Contribute to the Conversion of Ly6Chigh Monocytes into Ly6Clow Subsets under AMI.Stem Cells Int. 2020 Jan 13;2020:2460158. doi: 10.1155/2020/2460158. eCollection 2020. Stem Cells Int. 2020. PMID: 32399040 Free PMC article.
-
Guarana (Paullinia cupana) as a potential tool for mesenchymal stromal cells priming in regenerative medicine.Braz J Med Biol Res. 2024 Jul 29;57:e13286. doi: 10.1590/1414-431X2024e13286. eCollection 2024. Braz J Med Biol Res. 2024. PMID: 39082577 Free PMC article.
-
Advanced Nanomedicine Approaches for Myocardial Infarction Treatment.Int J Nanomedicine. 2024 Jun 24;19:6399-6425. doi: 10.2147/IJN.S467219. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38952676 Free PMC article. Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

