Evaluation of subcutaneous rituximab administration on Canadian systemic therapy suites
- PMID: 30464679
- PMCID: PMC6209563
- DOI: 10.3747/co.25.4231
Evaluation of subcutaneous rituximab administration on Canadian systemic therapy suites
Abstract
Background: Non-Hodgkin lymphoma (nhl) is the most common hematologic malignancy. Diffuse large B-cell lymphoma (dlbcl) and follicular lymphoma (fl) constitute 55% of new nhl cases and are initially treated with rituximab-based chemoimmunotherapy. Relative to intravenous (IV) rituximab, a subcutaneous (sc) formulation approved in 2016 has comparable pharmacokinetics, efficacy, and safety, and a greatly reduced administration time; it is also preferred by patients. The objective of the present study was to estimate the effect (on systemic therapy suite time and on the costs of drug acquisition and administration) of implementing sc rituximab in the initial chemoimmunotherapy for fl and dlbcl over 3 years in the Canadian market.
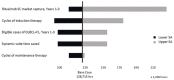
Methods: An Excel (Microsoft Corporation, Redmond, WA, U.S.A.)-based model was created with a population size based on epidemiologic data and current rituximab use, duration of use considering initial therapy, time savings for sc rituximab administration from published studies, costs from standard Canadian sources, and assumed uptake in implementing provinces of 65%, 75%, and 80% over 3 years. Key parameters and sensitivity analysis values were validated by clinical experts located in various Canadian jurisdictions. Costs are reported in 2017 Canadian dollars from the perspective of the health care system.
Results: More than 3 years after implementation of sc rituximab, we estimated that 5762 Canadians would be receiving sc rituximab, resulting in savings of 128,715 hours in systemic therapy suite time and approximately $40 million in drug and administration costs. Sensitivity analyses suggest that the model is most sensitive to sc market uptake, number of induction therapy cycles, and eligible patients.
Conclusions: Subcutaneous administration of rituximab can significantly reduce systemic therapy suite time and achieve substantial savings in drug and administration costs.
Keywords: Non-Hodgkin lymphoma; cost analyses; intravenous rituximab; rituximab; subcutaneous rituximab.
Conflict of interest statement
CONFLICT OF INTEREST DISCLOSURES We have read and understood Current Oncology’s policy on disclosing conflicts of interest, and we declare the following interests: DAS has served on ad hoc advisory boards for Roche, Janssen, Gilead, AbbVie, Celgene, Amgen, Lundbeck, Merck, Bristol–Myers Squibb, Seattle Genetics, and Servier; JSB has served on advisory boards for Janssen, Celgene, Amgen, Roche, and Takeda; BM and DB are Roche employees; RF has participated on ad hoc advisory boards for Celgene, Janssen, Novartis, and Gilead.
Figures

References
-
- Canadian Cancer Society. Canadian Cancer Statistics 2017. Toronto, ON: Canadian Cancer Society; 2017.
-
- Canadian Cancer Society. Non-Hodgkin Lymphoma Statistics [Web page] Toronto, ON: Canadian Cancer Society; 2017. [Available at: http://www.cancer.ca/en/cancer-information/cancer-type/non-hodgkin-lymph...; cited 17 January 2018]
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

