Glycoprotein D of HSV-1 is dependent on tegument protein UL16 for packaging and contains a motif that is differentially required for syncytia formation
- PMID: 30465930
- PMCID: PMC6400489
- DOI: 10.1016/j.virol.2018.09.018
Glycoprotein D of HSV-1 is dependent on tegument protein UL16 for packaging and contains a motif that is differentially required for syncytia formation
Abstract
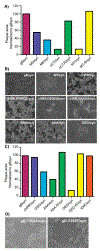
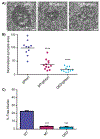
Glycoprotein D (gD) of herpes simplex virus type 1 (HSV-1) plays a key role in multiple events during infection including virus entry, cell-to-cell spread, and virus-induced syncytia formation. Here, we provide evidence that an arginine/lysine cluster located at the transmembrane-cytoplasm interface of gD critically contributes to viral spread and cell-cell fusion. Our studies began with the discovery that packaging of gD into virions is almost completely blocked in the absence of tegument protein UL16. We subsequently identified a novel, direct, and regulated interaction between UL16 and gD, but this was not important for syncytia formation. However, a mutational analysis of the membrane-proximal basic residues of gD revealed that they are needed for the gBsyn phenotype, salubrinal-induced fusion of HSV-infected cells, and cell-to-cell spread. Finally, we found that these same gD tail basic residues are not required for cell fusion induced by a gKsyn variant.
Keywords: Cell fusion, syncytia; Cell-to-cell spread; Glycoprotein D; Herpes simplex virus; Packaging; Protein interactions; Tegument; UL16.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures






References
-
- Ambrosini A, Enquist L, 2015. Cell-Fusion events induced by alpha-herpesviruses. Future Medicine, Future Virology, pp. 185–200.
-
- Balan P, Davis-Poynter N, Bell S, Atkinson H, Browne H, Minson T, 1994. An analysis of the in vitro and in vivo phenotypes of mutants of herpes simplex virus type 1 lacking glycoproteins gG, gE, gI or the putative gJ. J Gen Virol 75(Pt 6), 1245–1258. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

