Cost-effective production of tag-less recombinant protein in Nicotiana benthamiana
- PMID: 30468023
- PMCID: PMC6523591
- DOI: 10.1111/pbi.13040
Cost-effective production of tag-less recombinant protein in Nicotiana benthamiana
Abstract
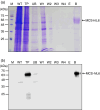
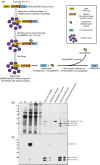
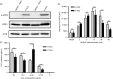
Plants have recently received a great deal of attention as a means of producing recombinant proteins. Despite this, a limited number of recombinant proteins are currently on the market and, if plants are to be more widely used, a cost-effective and efficient purification method is urgently needed. Although affinity tags are convenient tools for protein purification, the presence of a tag on the recombinant protein is undesirable for many applications. A cost-effective method of purification using an affinity tag and the removal of the tag after purification has been developed. The family 3 cellulose-binding domain (CBM3), which binds to microcrystalline cellulose, served as the affinity tag and the small ubiquitin-related modifier (SUMO) and SUMO-specific protease were used to remove it. This method, together with size-exclusion chromatography, enabled purification of human interleukin-6 (hIL6) with a yield of 18.49 mg/kg fresh weight from leaf extracts of Nicotiana benthamiana following Agrobacterium-mediated transient expression. Plant-produced hIL6 (P-hIL6) contained less than 0.2 EU/μg (0.02 ng/mL) endotoxin. P-hIL6 activated the Janus kinase-signal transducer and activator of transcriptional pathways in human LNCaP cells, and induced expression of IL-21 in activated mouse CD4+ T cells. This approach is thus a powerful method for producing recombinant proteins in plants.
Keywords: Nicotiana benthamiana; bdSENP1; bdSUMO; cellulose-binding domain; human interleukin-6; plant-based expression system; proteolytic tag removal.
© 2018 The Authors. Plant Biotechnology Journal published by Society for Experimental Biology and The Association of Applied Biologists and John Wiley & Sons Ltd.
Conflict of interest statement
The authors have no financial conflicts of interest to report.
Figures



Similar articles
-
In Vivo Removal of N-Terminal Fusion Domains From Recombinant Target Proteins Produced in Nicotiana benthamiana.Front Plant Sci. 2020 Apr 8;11:440. doi: 10.3389/fpls.2020.00440. eCollection 2020. Front Plant Sci. 2020. PMID: 32328082 Free PMC article.
-
Overexpression and Purification of Gracilariopsis chorda Carbonic Anhydrase (GcCAα3) in Nicotiana benthamiana, and Its Immobilization and Use in CO2 Hydration Reactions.Front Plant Sci. 2020 Nov 19;11:563721. doi: 10.3389/fpls.2020.563721. eCollection 2020. Front Plant Sci. 2020. PMID: 33329625 Free PMC article.
-
A Chimeric Affinity Tag for Efficient Expression and Chromatographic Purification of Heterologous Proteins from Plants.Front Plant Sci. 2016 Feb 15;7:141. doi: 10.3389/fpls.2016.00141. eCollection 2016. Front Plant Sci. 2016. PMID: 26913045 Free PMC article.
-
Molecular responses of agroinfiltrated Nicotiana benthamiana leaves expressing suppressor of silencing P19 and influenza virus-like particles.Plant Biotechnol J. 2024 May;22(5):1078-1100. doi: 10.1111/pbi.14247. Epub 2023 Dec 2. Plant Biotechnol J. 2024. PMID: 38041470 Free PMC article. Review.
-
Current strategies for the use of affinity tags and tag removal for the purification of recombinant proteins.Protein Expr Purif. 2006 Jul;48(1):1-13. doi: 10.1016/j.pep.2005.12.002. Epub 2005 Dec 28. Protein Expr Purif. 2006. PMID: 16427311 Review.
Cited by
-
Optimising expression and extraction of recombinant proteins in plants.Front Plant Sci. 2022 Dec 8;13:1074531. doi: 10.3389/fpls.2022.1074531. eCollection 2022. Front Plant Sci. 2022. PMID: 36570881 Free PMC article. Review.
-
Transient Expression of Dengue Virus NS1 Antigen in Nicotiana benthamiana for Use as a Diagnostic Antigen.Front Plant Sci. 2020 Jan 16;10:1674. doi: 10.3389/fpls.2019.01674. eCollection 2019. Front Plant Sci. 2020. PMID: 32010161 Free PMC article.
-
GREEN FLUORESCENT PROTEIN variants with enhanced folding are more efficiently imported into chloroplasts.Plant Physiol. 2022 Aug 29;190(1):238-249. doi: 10.1093/plphys/kiac291. Plant Physiol. 2022. PMID: 35699510 Free PMC article.
-
Recent advances in expression and purification strategies for plant made vaccines.Front Plant Sci. 2023 Nov 23;14:1273958. doi: 10.3389/fpls.2023.1273958. eCollection 2023. Front Plant Sci. 2023. PMID: 38078091 Free PMC article. Review.
-
Development of a tag-free plant-made interferon gamma production system with improved therapeutic efficacy against viruses.Front Bioeng Biotechnol. 2024 Jan 11;11:1341340. doi: 10.3389/fbioe.2023.1341340. eCollection 2023. Front Bioeng Biotechnol. 2024. PMID: 38274005 Free PMC article.
References
-
- Arnau, J. , Lauritzen, C. , Petersen, G.E. and Pedersen, J. (2006) Current strategies for the use of affinity tags and tag removal for the purification of recombinant proteins. Protein Expr. Purif. 48, 1–13. - PubMed
-
- Bayer, E.A. , Chanzy, H. , Lamed, R. and Shoham, Y. (1998) Cellulose, cellulases and cellulosomes. Proc. Natl Acad. Sci. USA, 8, 548–557. - PubMed
-
- Bereli, N. , Akgöl, S. , Yavuz, H. and Denizli, A. (2005) Antibody purification by concanavalin A affinity chromatography. J. Appl. Polym. Sci. 97, 1202–1208.
-
- Berlec, A. and Štrukelj, B. (2013) Current state and recent advances in biopharmaceutical production in Escherichia coli, yeasts and mammalian cells. J. Ind. Microbiol. Biotechnol. 40, 257–274. - PubMed
-
- Björck, L. and Kronvall, G. (1984) Purification and some properties of streptococcal protein G, a novel IgG‐binding reagent. J. Immunol. 133, 969–974. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

