Antibody-mediated immunosuppression can result from RBC antigen loss independent of Fcγ receptors in mice
- PMID: 30474857
- PMCID: PMC7145427
- DOI: 10.1111/trf.14939
Antibody-mediated immunosuppression can result from RBC antigen loss independent of Fcγ receptors in mice
Abstract
Background: Anti-RhD administration can prevent de novo anti-RhD formation following RhD+ red blood cell (RBC) exposure, termed antibody-mediated immunosuppression (AMIS). Recent studies suggest that AMIS may occur through target antigen alterations, known as antigen modulation. However, studies suggest that AMIS may occur independent of antigen modulation. In particular, AMIS to RBCs that transgenically express the fusion hen egg lysozyme-ovalbumin-Duffy (HOD) antigen have been shown to occur independent of activating Fcγ receptors (FcγRs) thought to be required for antigen modulation. Therefore, we sought to determine the mechanism behind AMIS following HOD RBC exposure.
Study design and methods: Following transfer of HOD RBCs into wild-type or FcγR-chain knockout recipients in the presence or absence of monoclonal anti-hen egg lysozyme (HEL) antibody, individually or in combination, HOD antigen levels and anti-HOD antibody formation were examined.
Results: Our results demonstrate that anti-HEL antibodies individually or in combination suppressed anti-HOD IgM, which correlated with the rate of detectable decrease in HEL on HOD RBCs. Furthermore, exposure to anti-HEL antibodies alone or in combination equally suppressed anti-HOD IgG formation. Unexpectedly, combination or individual anti-HEL antibodies induced AMIS and antigen modulation in an FcγR-independent manner. Pre-exposure of HOD RBCs to anti-HEL antibodies reduced antigen levels and suppressed anti-HOD antibody formation following HOD RBC exposure.
Conclusion: These results suggest that antibody-mediated antigen modulation may reflect a mechanism of AMIS that can occur independent of activating FcγRs and may provide a surrogate to identify antibodies capable of inducing AMIS against different RBC antigens.
© 2018 AABB.
Conflict of interest statement
CONFLICT OF INTEREST
The authors have disclosed no conflicts of interest.
Figures

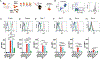

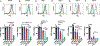
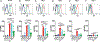

Similar articles
-
IgG-Mediated Immune Suppression to Erythrocytes by Polyclonal Antibodies Can Occur in the Absence of Activating or Inhibitory Fcγ Receptors in a Full Mouse Model.J Immunol. 2015 Sep 1;195(5):2224-30. doi: 10.4049/jimmunol.1500790. Epub 2015 Jul 17. J Immunol. 2015. PMID: 26188060
-
Antibody-mediated immune suppression by antigen modulation is antigen-specific.Blood Adv. 2018 Nov 13;2(21):2986-3000. doi: 10.1182/bloodadvances.2018018408. Blood Adv. 2018. PMID: 30413434 Free PMC article.
-
Antibody-mediated immune suppression of erythrocyte alloimmunization can occur independently from red cell clearance or epitope masking in a murine model.J Immunol. 2014 Sep 15;193(6):2902-10. doi: 10.4049/jimmunol.1302287. Epub 2014 Aug 13. J Immunol. 2014. PMID: 25122924
-
Mechanisms of anti-D action in the prevention of hemolytic disease of the fetus and newborn.Hematology Am Soc Hematol Educ Program. 2009:185-91. doi: 10.1182/asheducation-2009.1.185. Hematology Am Soc Hematol Educ Program. 2009. PMID: 20008198 Review.
-
Mechanisms of anti-D action in the prevention of hemolytic disease of the fetus and newborn: what can we learn from rodent models?Curr Opin Hematol. 2009 Nov;16(6):488-96. doi: 10.1097/MOH.0b013e32833199ed. Curr Opin Hematol. 2009. PMID: 19730101 Review.
Cited by
-
FcγRIV is required for IgG2c mediated enhancement of RBC alloimmunization.Front Immunol. 2022 Sep 13;13:972723. doi: 10.3389/fimmu.2022.972723. eCollection 2022. Front Immunol. 2022. PMID: 36189253 Free PMC article.
-
Examining the Role of Complement in Predicting, Preventing, and Treating Hemolytic Transfusion Reactions.Transfus Med Rev. 2019 Oct;33(4):217-224. doi: 10.1016/j.tmrv.2019.09.006. Epub 2019 Oct 18. Transfus Med Rev. 2019. PMID: 31679762 Free PMC article. Review.
-
Evaluation of the Bactericidal Activity of Galectins.Methods Mol Biol. 2022;2442:517-531. doi: 10.1007/978-1-0716-2055-7_27. Methods Mol Biol. 2022. PMID: 35320543
-
IgG Suppresses Antibody Responses to Sheep Red Blood Cells in Double Knock-Out Mice Lacking Complement Factor C3 and Activating Fcγ-Receptors.Front Immunol. 2020 Jul 8;11:1404. doi: 10.3389/fimmu.2020.01404. eCollection 2020. Front Immunol. 2020. PMID: 32733467 Free PMC article.
-
ABO blood group antigens and differential glycan expression: Perspective on the evolution of common human enzyme deficiencies.iScience. 2022 Dec 14;26(1):105798. doi: 10.1016/j.isci.2022.105798. eCollection 2023 Jan 20. iScience. 2022. PMID: 36691627 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

