Eicosanoid and Specialized Proresolving Mediator Regulation of Lymphoid Cells
- PMID: 30477730
- PMCID: PMC6379137
- DOI: 10.1016/j.tibs.2018.10.007
Eicosanoid and Specialized Proresolving Mediator Regulation of Lymphoid Cells
Abstract
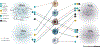
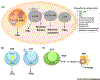
Eicosanoids and specialized proresolving mediators (SPMs) regulate leukocyte function and inflammation. They are ideally positioned at the interface of the innate and adaptive immune responses when lymphocytes interact with leukocytes. Receptors for leukotriene B4 (LTB4), prostaglandin E2 (PGE2), and SPMs are expressed on lymphocytes. Evidence points toward an essential role of these lipid mediators (LMs) in direct regulation of lymphocyte functions. SPMs, which include lipoxins, demonstrate comprehensive protective actions with lymphocytes. LTB4 and PGE2 regulation of lymphocytes is diverse and depends on the interaction of lymphocytes with other cells. Importantly, both LTB4 and PGE2 are essential regulators of T cell antitumor activity. These LMs are attractive therapeutic targets to control dysregulated innate and adaptive immune responses, promote lymphocyte antitumor activity, and prevent tumor immune evasion.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Eicosanoid pathway on host resistance and inflammation during Mycobacterium tuberculosis infection is comprised by LTB4 reduction but not PGE2 increment.Biochim Biophys Acta Mol Basis Dis. 2020 Mar 1;1866(3):165574. doi: 10.1016/j.bbadis.2019.165574. Epub 2019 Oct 28. Biochim Biophys Acta Mol Basis Dis. 2020. PMID: 31666208
-
In vivo rectal inflammatory mediator changes with radiotherapy to the pelvis.Gut. 1993 Sep;34(9):1210-4. doi: 10.1136/gut.34.9.1210. Gut. 1993. PMID: 8406156 Free PMC article.
-
Eicosanoids in skin of patients with atopic dermatitis: prostaglandin E2 and leukotriene B4 are present in biologically active concentrations.J Allergy Clin Immunol. 1989 Feb;83(2 Pt 1):450-5. doi: 10.1016/0091-6749(89)90132-2. J Allergy Clin Immunol. 1989. PMID: 2537352
-
Metabololipidomic profiling of functional immunoresolvent clusters and eicosanoids in mammalian tissues.Biochem Biophys Res Commun. 2018 Oct 7;504(3):553-561. doi: 10.1016/j.bbrc.2018.03.037. Epub 2018 Mar 15. Biochem Biophys Res Commun. 2018. PMID: 29524409 Free PMC article. Review.
-
Crosstalk between purinergic receptors and lipid mediators in leishmaniasis.Parasit Vectors. 2016 Sep 5;9(1):489. doi: 10.1186/s13071-016-1781-1. Parasit Vectors. 2016. PMID: 27595742 Free PMC article. Review.
Cited by
-
Lipidomic Profiling of Bronchoalveolar Lavage Fluid Extracellular Vesicles Indicates Their Involvement in Lipopolysaccharide-Induced Acute Lung Injury.J Innate Immun. 2022;14(5):555-568. doi: 10.1159/000522338. Epub 2022 Apr 1. J Innate Immun. 2022. PMID: 35367992 Free PMC article.
-
Arachidonic acid in aging: New roles for old players.J Adv Res. 2025 Apr;70:79-101. doi: 10.1016/j.jare.2024.05.003. Epub 2024 May 4. J Adv Res. 2025. PMID: 38710468 Free PMC article. Review.
-
Redressing the interactions between stem cells and immune system in tissue regeneration.Biol Direct. 2021 Oct 20;16(1):18. doi: 10.1186/s13062-021-00306-6. Biol Direct. 2021. PMID: 34670590 Free PMC article. Review.
-
Regulation of Diseases-Associated Microglia in the Optic Nerve by Lipoxin B4 and Ocular Hypertension.bioRxiv [Preprint]. 2024 Oct 28:2024.03.18.585452. doi: 10.1101/2024.03.18.585452. bioRxiv. 2024. Update in: Mol Neurodegener. 2024 Nov 20;19(1):86. doi: 10.1186/s13024-024-00775-z. PMID: 38562864 Free PMC article. Updated. Preprint.
-
A Role for Lipid Mediators in Acute Myeloid Leukemia.Int J Mol Sci. 2019 May 16;20(10):2425. doi: 10.3390/ijms20102425. Int J Mol Sci. 2019. PMID: 31100828 Free PMC article. Review.

