Overlooked Issues on Pharmacokinetics Data Interpretation of Protein Drugs-a Case Example of Erythropoietin
- PMID: 30478521
- PMCID: PMC7472663
- DOI: 10.1208/s12248-018-0269-7
Overlooked Issues on Pharmacokinetics Data Interpretation of Protein Drugs-a Case Example of Erythropoietin
Keywords: erythropoietin; overlooked issues; pharmacokinetic data interpretation; protein drugs.
Figures
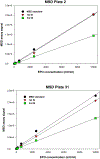
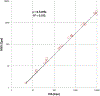
Similar articles
-
An overview of the pharmacokinetic disposition of darbepoetin alfa.Pharmacotherapy. 2002 Sep;22(9 Pt 2):133S-140S. doi: 10.1592/phco.22.14.133s.33396. Pharmacotherapy. 2002. PMID: 12222583 Review.
-
Control-relevant erythropoiesis modeling in end-stage renal disease.IEEE Trans Biomed Eng. 2014 Mar;61(3):658-64. doi: 10.1109/TBME.2013.2286100. Epub 2013 Oct 16. IEEE Trans Biomed Eng. 2014. PMID: 24235247
-
Erythropoietin stimulating agents and epoetin alfa revisited: what's really relevant?Clin J Am Soc Nephrol. 2008 Jul;3(4):935-7. doi: 10.2215/CJN.02300508. Epub 2008 Jun 25. Clin J Am Soc Nephrol. 2008. PMID: 18579671 No abstract available.
-
[Current issues in erythropoietin therapy of renal anemia].Lege Artis Med. 2007 Oct;17(10):667-73. Lege Artis Med. 2007. PMID: 19227596 Review. Hungarian.
-
Population pharmacokinetics meta-analysis of recombinant human erythropoietin in healthy subjects.Clin Pharmacokinet. 2007;46(2):159-73. doi: 10.2165/00003088-200746020-00004. Clin Pharmacokinet. 2007. PMID: 17253886
Cited by
-
Target-mediated disposition population pharmacokinetics model of erythropoietin in premature neonates following multiple intravenous and subcutaneous dosing regimens.Eur J Pharm Sci. 2019 Oct 1;138:105013. doi: 10.1016/j.ejps.2019.105013. Epub 2019 Jul 21. Eur J Pharm Sci. 2019. PMID: 31340188 Free PMC article. Clinical Trial.
References
-
- Storring PL, Gaines Das RE. The International Standard for Recombinant DNA-derived Erythropoietin: collaborative study of four recombinant DNA-derived erythropoietins and two highly purified human urinary erythropoietins. The Journal of endocrinology. 1992;134(3):459–84. - PubMed
-
- Wognum A Erythropoietin mini-review. https://wwwstemcellcom/erythropoietin-lphtml. 2015.
-
- NIBSC. Third WHO International Standard for Erythropoietin, recombinant, for bioassay NIBSC code 11/170. 2012:version 1.0.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

