Atomic Force Microscopy Analysis of EPPS-Driven Degradation and Reformation of Amyloid-β Aggregates
- PMID: 30480247
- PMCID: PMC6159726
- DOI: 10.3233/ADR-170024
Atomic Force Microscopy Analysis of EPPS-Driven Degradation and Reformation of Amyloid-β Aggregates
Abstract
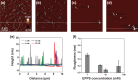
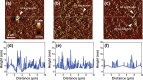
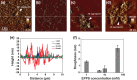
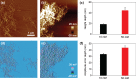
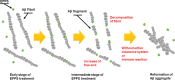
Amyloid-β (Aβ) peptides can be aggregated into β-sheet rich fibrils or plaques and deposited on the extracellular matrix of brain tissues, which is a hallmark of Alzheimer's disease. Several drug candidates have been designed to retard the progression of the neurodegenerative disorder or to eliminate toxic Aβ aggregates. Recently, 4-(2-Hydroxyethyl)-1-piperazinepropanesulfonic acid (EPPS) has emerged as a promising drug candidates for elimination of toxic Aβ aggregates. However, the effect of EPPS on the degradation of Aβ aggregates such as fibrils has not yet been fully elucidated. In this article, we investigate the EPPS-driven degradative behavior of Aβ aggregates at the molecular level by using high-resolution atomic force microscopy. We synthesized Aβ fibrils and observed degradation of fibrils following treatment with various concentrations (1-50 mM) of EPPS for various time periods. We found that degradation of Aβ fibrils by EPPS increased as a function of concentration and treatment duration. Intriguingly, we also found regeneration of Aβ aggregates with larger sizes than original aggregates at high concentrations (10 and 50 mM) of EPPS. This might be attributed to a shorter lag phase that facilitates reformation of Aβ aggregates in the absence of clearance system.
Keywords: 4-(2-Hydroxyethyl)-1-piperazinepropanesulfonic acid (EPPS); Alzheimer’s disease; Aβ aggregates; amyloid-β; atomic force microscopy; fibril degradation.
Figures


Similar articles
-
Deciphering the Disaggregation Mechanism of Amyloid Beta Aggregate by 4-(2-Hydroxyethyl)-1-Piperazinepropanesulfonic Acid Using Electrochemical Impedance Spectroscopy.Sensors (Basel). 2021 Jan 25;21(3):788. doi: 10.3390/s21030788. Sensors (Basel). 2021. PMID: 33503934 Free PMC article.
-
Chemical-Driven Amyloid Clearance for Therapeutics and Diagnostics of Alzheimer's Disease.Acc Chem Res. 2024 Nov 19;57(22):3266-3276. doi: 10.1021/acs.accounts.4c00458. Epub 2024 Nov 4. Acc Chem Res. 2024. PMID: 39496112
-
Light-triggered dissociation of self-assembled β-amyloid aggregates into small, nontoxic fragments by ruthenium (II) complex.Acta Biomater. 2018 Feb;67:147-155. doi: 10.1016/j.actbio.2017.11.048. Epub 2017 Dec 6. Acta Biomater. 2018. PMID: 29221856
-
Amyloid-beta aggregates formed at polar-nonpolar interfaces differ from amyloid-beta protofibrils produced in aqueous buffers.Microsc Res Tech. 2005 Jul;67(3-4):164-74. doi: 10.1002/jemt.20189. Microsc Res Tech. 2005. PMID: 16103999 Review.
-
Elucidating the Structures of Amyloid Oligomers with Macrocyclic β-Hairpin Peptides: Insights into Alzheimer's Disease and Other Amyloid Diseases.Acc Chem Res. 2018 Mar 20;51(3):706-718. doi: 10.1021/acs.accounts.7b00554. Epub 2018 Mar 6. Acc Chem Res. 2018. PMID: 29508987 Free PMC article. Review.
Cited by
-
Differential Clearance of Aβ Species from the Brain by Brain Lymphatic Endothelial Cells in Zebrafish.Int J Mol Sci. 2021 Nov 2;22(21):11883. doi: 10.3390/ijms222111883. Int J Mol Sci. 2021. PMID: 34769316 Free PMC article.
-
Amyloid Formation in Nanoliter Droplets.Int J Mol Sci. 2022 May 13;23(10):5480. doi: 10.3390/ijms23105480. Int J Mol Sci. 2022. PMID: 35628295 Free PMC article.
-
Melanoma Detection by AFM Indentation of Histological Specimens.Diagnostics (Basel). 2022 Jul 17;12(7):1736. doi: 10.3390/diagnostics12071736. Diagnostics (Basel). 2022. PMID: 35885640 Free PMC article.
-
Interactions between Curcumin Derivatives and Amyloid-β Fibrils: Insights from Molecular Dynamics Simulations.J Chem Inf Model. 2020 Jan 27;60(1):289-305. doi: 10.1021/acs.jcim.9b00561. Epub 2019 Dec 20. J Chem Inf Model. 2020. PMID: 31809572 Free PMC article.
References
-
- Dobson CM (1999) Protein misfolding, evolution and disease. Trends Biochem Sci 24, 329–332. - PubMed
-
- Merlini G, Bellotti V (2003) Molecular mechanisms of amyloidosis. New Engl J Med 349, 583–596. - PubMed
-
- Blennow K, Hampel H, Weiner M, Zetterberg H (2010) Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nat Rev Neurol 6, 131–144. - PubMed
-
- Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 297, 353–356. - PubMed
-
- Bayliss D, Walsh JL, Shama G, Iza F, Kong MG (2009) Reduction and degradation of amyloid aggregates by a pulsed radio-frequency cold atmospheric plasma jet. New J Phys 11, 115024.
LinkOut - more resources
Full Text Sources

