Corticosteroids for septic arthritis in children
- PMID: 30480764
- PMCID: PMC6517045
- DOI: 10.1002/14651858.CD012125.pub2
Corticosteroids for septic arthritis in children
Abstract
Background: Septic arthritis is an acute infection of the joints characterised by erosive disruption of the articular space. It is the most common non-degenerative articular disease in developing countries. The most vulnerable population for septic arthritis includes infants and preschoolers, especially boys. Septic arthritis disproportionately affects populations of low socioeconomic status. Systemic corticosteroids and antibiotic therapy may be beneficial for treatment of septic arthritis. Even if the joint infection is eradicated by antibiotic treatment, the inflammatory process may produce residual joint damage and sequelae.
Objectives: To determine the benefits and harms of corticosteroids as adjunctive therapy in children with a diagnosis of septic arthritis.
Search methods: We searched MEDLINE, Embase, the Cochrane Central Register of Controlled Trials (CENTRAL), in the Cochrane Library, Latin American Caribbean Health Sciences Literature (LILACS), the World Health Organization (WHO) trials portal (www.who.int/ictrp/en/), ClinicalTrials.gov (www.ClinicalTrials.gov), and Google Scholar. We searched all databases from their inception to 17 April 2018, with no restrictions on language of publication.
Selection criteria: We included randomised controlled trials (RCTs) with patients from two months to 18 years of age with a diagnosis of septic arthritis who were receiving corticosteroids in addition to antibiotic therapy or as an adjuvant to other therapies such as surgical drainage, intra-articular puncture, arthroscopic irrigation, or debridement.
Data collection and analysis: Two review authors independently assessed eligibility, data extraction, and evaluation of risk of bias. We considered as major outcomes the presence of pain, activities of daily living, normal physical joint function, days of antibiotic treatment, length of hospital stay, and numbers of total and serious adverse events. We used standard methodological procedures expected by Cochrane. We assessed the evidence using GRADE (Grading of Recommendations Assessment, Development and Evaluation) and created a 'Summary of findings' table.
Main results: We included two RCTs involving a total of 149 children between three months and 18 years of age who were receiving antibiotics for septic arthritis. The most commonly affected joints were hips and knees. These studies were performed in Costa Rica and Israel. In both studies, dexamethasone administered intravenously (ranging from 0.15 to 0.2 mg/kg/dose every six to eight hours) during four days was the corticosteroid, and the comparator was placebo. Trials excluded patients with any degree of immunodeficiency or immunosuppression. The longest follow-up was one year. Trials did not report activities of daily living nor length of hospital stay. Both studies used adequate processes for randomisation, allocation concealment, and blinding, and review authors judged them to have low risk of selection and performance bias. Losses to follow-up were substantive in both studies, and we judged them to have high risk of attrition bias and of selective outcome reporting. We graded all outcomes as low quality due to concerns about study limitations and imprecision.The risk ratio (RR) for absence of pain at 12 months of follow-up was 1.33, favouring corticosteroids (95% confidence interval (CI) 1.03 to 1.72; P = 0.03; number needed to treat for an additional beneficial outcome (NNTB) = 13, 95% CI 6 to 139; absolute risk difference 24%, 95% CI 5% to 43%).The RR for normal function of the affected joint at 12 months of follow-up was 1.32, favouring corticosteroids (95% CI 1.12 to 1.57; P = 0.001; NNTB = 13, 95% CI 7 to 33; absolute risk difference 24%, 95% CI 11% to 37%).We found a reduction in the number of days of intravenous antibiotic treatment favouring corticosteroids (mean difference (MD) -2.77, 95% CI -4.16 to -1.39) based on two trials with 149 participants.Researchers did not report length of hospital stay. One trial (49 participants) reported that treatment with dexamethasone was associated with a shorter duration of IV antibiotic treatment, leading to a shorter hospital stay, and although duration of hospitalisation was a primary outcome of the study, study authors did not provide data on the duration of hospitalisation. We downgraded the quality by one level for concerns about study limitations (high risk of attrition bias and selective reporting), and by another level for imprecision.In one trial of 49 participants, researchers followed 29 children for 12 months, and parents reported that no children demonstrated adverse effects of the intervention.
Authors' conclusions: Evidence for corticosteroids as adjunctive therapy in children with a diagnosis of septic arthritis is of low quality and is derived from the findings of two trials (N = 149). Corticosteroids may increase the proportion of patients without pain and the proportion of patients with normal function of the affected joint at 12 months, and may also reduce the number of days of antibiotic treatment. However, we cannot draw strong conclusions based upon these trial results. Additional randomised clinical trials in children with relevant outcomes are needed.
Conflict of interest statement
None declared.
Figures
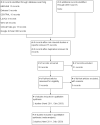

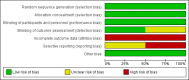










Comment in
-
Are corticosteroids effective as adjunctive therapy in children with septic arthritis? A Cochrane Review summary with commentary.Int J Rheum Dis. 2020 Oct;23(10):1426-1429. doi: 10.1111/1756-185X.13903. Int J Rheum Dis. 2020. PMID: 33104295 No abstract available.
References
References to studies included in this review
Harel 2011 {published data only}
-
- Harel L, Prais D, Bar‐On E, Livni G, Hoffer V, Uziel Y, Amir J. Dexamethasone therapy for septic arthritis in children: results of a randomized double‐blind placebo‐controlled study. Journal of Pediatric Orthopaedics 2011;31(2):211‐5. [PUBMED: 21307717] - PubMed
Odio 2003 {published data only}
-
- Odio CM, Ramirez T, Arias G, Abdelnour A, Hidalgo I, Herrera ML, et al. Double blind, randomized, placebo‐controlled study of dexamethasone therapy for hematogenous septic arthritis in children. Pediatric Infectious Diseases Journal 2003;22(10):883‐8. [14551489] - PubMed
References to studies excluded from this review
Arti 2014 {published data only}
Fogel 2015 {published data only}
-
- Fogel I, Amir J, Bar‐On E, Harel L. Dexamethasone therapy for septic arthritis in children. Pediatrics 2015;136(4):e776‐82. [PUBMED: 26347429] - PubMed
van Oosterhout 2006 {published data only}
-
- Oosterhout M, Sont JK, Bajema IM, Breedveld FC, Laar JM. Comparison of efficacy of arthroscopic lavage plus administration of corticosteroids, arthroscopic lavage plus administration of placebo, and joint aspiration plus administration of corticosteroids in arthritis of the knee: a randomized controlled trial. Arthritis & Rheumatism 2006;55(6):964‐70. [PUBMED: 17139644] - PubMed
Additional references
Al Saadi 2009
-
- Al Saadi MM, Al Zamil FA, Bokhary NA, Al Shamsan LA, Al Alola SA, Al Eissa YS. Acute septic arthritis in children. Pediatrics International 2009;51(3):377‐80. - PubMed
Aljeab 2016
Annane 2009
Baghdadi 2012
-
- Baghdadi T, Saberi S, Sobhani Eraghi A, Arabzadeh A, Mardookhpour S. Late sequelae of hip septic arthritis in children. Acta Medica Iranica 2012;50(7):463‐7. - PubMed
Bremell 1992
Brouwer 2015
Cates 2008 [Computer program]
-
- Christopher Cates EBM website. http://www.nntonline.net. Visual Rx. Version 3. Christopher Cates EBM website. http://www.nntonline.net, 2008.
Choe 2013
-
- Choe H, Inaba Y, Kobayashi N, Aoki C, Machida J, Nakamura N, et al. Use of real‐time polymerase chain reaction for the diagnosis of infection and differentiation between gram‐positive and gram‐negative septic arthritis in children. Journal of Pediatric Orthopedics 2013;33(3):e28‐33. - PubMed
Colavite 2014
Dodwell 2013
-
- Dodwell ER. Osteomyelitis and septic arthritis in children: current concepts. Current Opinion in Pediatrics 2013;25(1):58‐63. - PubMed
Farrow 2015
Forero 2013
-
- Forero JM, Franco‐Moreno A, Delgado‐Noguera M. Corticosteroids for septic arthritis in children [Corticoesteroides en artritis séptica en niños]. Revista de la Facultad de Ciencias de la Salud Universidad del Cauca 2013;15(2):26‐9.
García‐Arias 2011
-
- García‐Arias M, Balsa A, Mola EM. Septic arthritis. Best Practice & Research. Clinical Rheumatology 2011;25(3):407‐21. - PubMed
Ghogomu 2014
-
- Ghogomu EA, Maxwell LJ, Buchbinder R, Rader T, Pardo Pardo J, Johnston RV, et al. Updated method guidelines for Cochrane musculoskeletal group systematic reviews and metaanalyses. Journal of Rheumatology 2014;41(2):194‐205. - PubMed
GRADEpro 2015 [Computer program]
-
- McMaster University. GRADEpro GDT: GRADEpro Guideline Development Tool. Evidence Prime, 2015.
Head 2016
Higgins 2011
-
- Higgins JPT, Altman DG, Sterne JAC (editors). Chapter 8 ‐ Assessing risk of bias in included studies. In: Higgins JPT, Green S (editors), Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Kang 2009
-
- Kang SN, Sanghera T, Mangwani J, Paterson JMH, Ramachandran M. The management of septic arthritis in children: systematic review of the English language literature. Journal of Bone and Joint Surgery 2009;91(9):1127‐33. - PubMed
Kwan 2004
-
- Kwan Tat S, Padrines M, Theoleyre S, Heymann D, Fortun Y. IL‐6, RANKL,TNF‐alpha/IL‐1: interrelations in bone resorption pathophysiology. Cytokine & Growth Factor Reviews 2004;15:49‐60. - PubMed
Mathews 2008a
-
- Mathews CJ, Coakley G. Septic arthritis: current diagnostic and therapeutic algorithm. Current Opinion in Rheumatology 2008;20(4):457‐62. - PubMed
Mathews 2008b
-
- Mathews CJ, Kingsley G, Field M, Jones A, Weston VC, Phillips M, et al. Management of septic arthritis: a systematic review. Postgraduate Medical Journal 2008;84(991):265‐70. - PubMed
Mathews 2010
-
- Mathews CJ, Weston VC, Jones A, Field M, Coakley G. Bacterial septic arthritis in adults. Lancet 2010;375(9717):846‐55. - PubMed
Ogunlese 2015
RevMan 2014 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schünemann 2011
-
- Schünemann HJ, Oxman AD, Higgins JPT, Vist GE, Glasziou P, Guyatt GH. Chapter 11 ‐ Presenting results and ‘Summary of findings' tables. In: Higgins JPT, Green S (editors), Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Shirtliff 2002
Sterne 2011
-
- Sterne JAC, Egger M, Moher D (editors). Chapter 10 ‐ Addressing reporting biases. In Higgins JPT, Green S (editors), Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Young 2011
-
- Young TP, Maas L, Thorp AW, Brown L. Etiology of septic arthritis in children: an update for the new millennium. American Journal of Emergency Medicine 2011;29(8):899‐902. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

