Physiological Studies of Chlorobiaceae Suggest that Bacillithiol Derivatives Are the Most Widespread Thiols in Bacteria
- PMID: 30482829
- PMCID: PMC6282198
- DOI: 10.1128/mBio.01603-18
Physiological Studies of Chlorobiaceae Suggest that Bacillithiol Derivatives Are the Most Widespread Thiols in Bacteria
Abstract
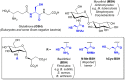
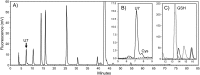
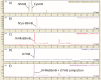
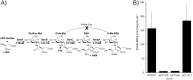
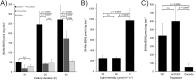
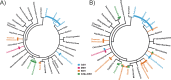
Low-molecular-weight (LMW) thiols mediate redox homeostasis and the detoxification of chemical stressors. Despite their essential functions, the distribution of LMW thiols across cellular life has not yet been defined. LMW thiols are also thought to play a central role in sulfur oxidation pathways in phototrophic bacteria, including the Chlorobiaceae Here we show that Chlorobaculum tepidum synthesizes a novel LMW thiol with a mass of 412 ± 1 Da corresponding to a molecular formula of C14H24N2O10S, which suggests that the new LMW thiol is closely related to bacillithiol (BSH), the major LMW thiol of low-G+C Gram-positive bacteria. The Cba. tepidum LMW thiol structure was N-methyl-bacillithiol (N-Me-BSH), methylated on the cysteine nitrogen, the fourth instance of this modification in metabolism. Orthologs of bacillithiol biosynthetic genes in the Cba. tepidum genome and the CT1040 gene product, N-Me-BSH synthase, were required for N-Me-BSH synthesis. N-Me-BSH was found in all Chlorobiaceae examined as well as Polaribacter sp. strain MED152, a member of the Bacteroidetes A comparative genomic analysis indicated that BSH/N-Me-BSH is synthesized not only by members of the Chlorobiaceae, Bacteroidetes, Deinococcus-Thermus, and Firmicutes but also by Acidobacteria, Chlamydiae, Gemmatimonadetes, and Proteobacteria. Thus, BSH and derivatives appear to be the most broadly distributed LMW thiols in biology.IMPORTANCE Low-molecular-weight thiols are key metabolites that participate in many basic cellular processes: central metabolism, detoxification, and oxidative stress resistance. Here we describe a new thiol, N-methyl-bacillithiol, found in an anaerobic phototrophic bacterium and identify a gene that is responsible for its synthesis from bacillithiol, the main thiol metabolite in many Gram-positive bacteria. We show that the presence or absence of this gene in a sequenced genome accurately predicts thiol content in distantly related bacteria. On the basis of these results, we analyzed genome data and predict that bacillithiol and its derivatives are the most widely distributed thiol metabolites in biology.
Keywords: Chlorobaculum tepidum; cellular redox status; chlorobiaceae; low molecular weight thiol; sulfur.
Copyright © 2018 Hiras et al.
Figures
Comment in
-
N-methyl-bacillithiol, a Novel Thiol from Anaerobic Bacteria.mBio. 2019 Jan 15;10(1):e02634-18. doi: 10.1128/mBio.02634-18. mBio. 2019. PMID: 30647158 Free PMC article.
Similar articles
-
N-methyl-bacillithiol, a Novel Thiol from Anaerobic Bacteria.mBio. 2019 Jan 15;10(1):e02634-18. doi: 10.1128/mBio.02634-18. mBio. 2019. PMID: 30647158 Free PMC article.
-
Biophysical features of bacillithiol, the glutathione surrogate of Bacillus subtilis and other firmicutes.Chembiochem. 2013 Nov 4;14(16):2160-8. doi: 10.1002/cbic.201300404. Epub 2013 Oct 2. Chembiochem. 2013. PMID: 24115506 Free PMC article.
-
Physiological roles of bacillithiol in intracellular metal processing.Curr Genet. 2016 Feb;62(1):59-65. doi: 10.1007/s00294-015-0511-0. Epub 2015 Aug 11. Curr Genet. 2016. PMID: 26259870 Review.
-
Application of genetically encoded redox biosensors to measure dynamic changes in the glutathione, bacillithiol and mycothiol redox potentials in pathogenic bacteria.Free Radic Biol Med. 2018 Nov 20;128:84-96. doi: 10.1016/j.freeradbiomed.2018.02.018. Epub 2018 Feb 15. Free Radic Biol Med. 2018. PMID: 29454879 Review.
-
Biosynthesis and functions of bacillithiol, a major low-molecular-weight thiol in Bacilli.Proc Natl Acad Sci U S A. 2010 Apr 6;107(14):6482-6. doi: 10.1073/pnas.1000928107. Epub 2010 Mar 22. Proc Natl Acad Sci U S A. 2010. PMID: 20308541 Free PMC article.
Cited by
-
The Crystal Structures of Bacillithiol Disulfide Reductase Bdr (YpdA) Provide Structural and Functional Insight into a New Type of FAD-Containing NADPH-Dependent Oxidoreductase.Biochemistry. 2020 Dec 29;59(51):4793-4798. doi: 10.1021/acs.biochem.0c00745. Epub 2020 Dec 16. Biochemistry. 2020. PMID: 33326741 Free PMC article.
-
The role of thiols in antioxidant systems.Free Radic Biol Med. 2019 Aug 20;140:14-27. doi: 10.1016/j.freeradbiomed.2019.05.035. Epub 2019 Jun 13. Free Radic Biol Med. 2019. PMID: 31201851 Free PMC article. Review.
-
The Bacillus subtilis monothiol bacilliredoxin BrxC (YtxJ) and the Bdr (YpdA) disulfide reductase reduce S-bacillithiolated proteins.Redox Biol. 2021 Jun;42:101935. doi: 10.1016/j.redox.2021.101935. Epub 2021 Mar 6. Redox Biol. 2021. PMID: 33722570 Free PMC article.
-
Staphylococcus aureus Uses the Bacilliredoxin (BrxAB)/Bacillithiol Disulfide Reductase (YpdA) Redox Pathway to Defend Against Oxidative Stress Under Infections.Front Microbiol. 2019 Jun 18;10:1355. doi: 10.3389/fmicb.2019.01355. eCollection 2019. Front Microbiol. 2019. PMID: 31275277 Free PMC article.
-
Surface-coated paper packaging with nanocellulose modified with quaternary ammonium organosilane and precipitated calcium carbonate for improved water repellency and antibacterial activity.Sci Rep. 2025 Jul 16;15(1):25723. doi: 10.1038/s41598-025-10306-5. Sci Rep. 2025. PMID: 40670488 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

