Differential characteristics in drug-induced autoimmune hepatitis
- PMID: 30483571
- PMCID: PMC6207017
- DOI: 10.1002/jgh3.12054
Differential characteristics in drug-induced autoimmune hepatitis
Abstract
Background and aim: Drug-induced autoimmune hepatitis (DIAIH) is an adverse effect associated with several drugs that usually occurs acutely, with variable latency, and it may potentially be mortal. There are a few reports and studies about DIAIH.
Methods: This was an analytical study of a retrospective cohort of patients, discriminated according to idiopathic or drug-induced etiology, followed up for a 7-year period until 31 December 2016.
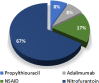
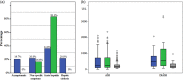
Results: A total of 190 patients were selected for the analysis, 12 (6.3%) with DIAIH. The two main drugs related to DIAIH were nitrofurantoin, n = 8 (67%), and NSAID, n = 2 (17%), constituting 84% of the cases. There were no significant differences in seropositivity between AIH with DIAIH in antinuclear antibodies (ANA) and anti-smooth muscle antibodies (ASMA) antibodies, with 82.6% versus 82.6% and 34% versus 16%, respectively. The fibrosis stages were similar, except for the F4 stage, in a greater proportion in AIH. None of the patients with DIAIH had cirrhosis or developed it during follow-up, but it was present in 42.1% of the AIH cases at diagnosis (P = 0.003). Biochemical remission with management was higher in DIAIH but not significant (91.7% vs 80.9%, P = 0.35). The definitive interruption of immunosuppression was successfully performed in 25% of those with DIAIH without relapses but was only possible in 2.8% in AIH (P < 0.001) with 32 cases of relapses.
Conclusion: DIAIH constitutes a minor proportion of AIH. The clinical and histological characteristics may be similar; DIAIH patients have a greater chance of having treatment suspended with a low risk of relapse, progression to cirrhosis, or need for liver transplant.
Keywords: autoimmune hepatitis; autoimmunity; drug‐induced liver injury; immunosuppression; prognosis.
Figures
 , No;
, No;  , Yes
, Yes , ALT P = 0.37;
, ALT P = 0.37;  , AST P = 0.37;
, AST P = 0.37;  , ALP P = 0.72.
, ALP P = 0.72.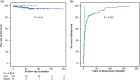
 , No;
, No;  , Yes.
, Yes.References
-
- Liu Z, Kaplowitz N. Immune‐mediated drug‐induced liver disease. Clin. Liver Dis. 2002; 6: 755–74. - PubMed
-
- Obermayer‐straub P, Strassburg CP, Manns MP, Strassburg CP, Target MPM. Target proteins in human autoimmunity: cytochromes glucuronosyltransferases. Can. J. Gastroenterol. 2000; 14: 429–39. - PubMed
-
- Hisamochi A, Kage M, Ide T et al An analysis of drug‐induced liver injury, which showed histological findings similar to autoimmune hepatitis. J. Gastroenterol. 2016; 51: 597–607. - PubMed
-
- Weiler‐Normann C, Schramm C. Drug induced liver injury and its relationship to autoimmune hepatitis. J Hepatol. 2011; 55: 747–9. - PubMed
LinkOut - more resources
Full Text Sources

