Expression of FKBP52 in the ovaries of PCOS rats
- PMID: 30483787
- PMCID: PMC6317667
- DOI: 10.3892/ijmm.2018.3998
Expression of FKBP52 in the ovaries of PCOS rats
Abstract
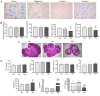
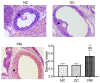
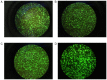
The present study aimed to examine the expression of FK‑506 binding protein 52 (FKBP52) in the ovary tissues of rats with polycystic ovarian syndrome (PCOS) and its action on mediating androgen receptor (AR) through the mitogen‑activated protein kinase (MAPK)/extracellular signal‑regulated kinase (ERK) pathway. PCOS model rats were established by dehydroepiandrosterone injection. Enzyme‑linked immunosorbent assay (ELISA) measured serum sex hormones. Hematoxylin and eosin (H&E) staining was used to examine histological changes of the ovarian tissues. The expression levels of FKBP52 were detected by immunohistochemical (IHC) staining, reverse transcription‑quantitative polymerase chain reaction (RT‑qPCR) analysis and western blotting (WB). In addition, RT‑qPCR analysis was used to detect the mRNA expression of AR, and WB was used to detect the protein expression levels of AR, ERK1/2 and phosphorylated (p‑)ERK1/2. In granulosa cell (GC) experiments, primary GCs were extracted and cultured. FKBP4 is the FKBP52‑encoding gene, therefore, adenovirus vectors Ad‑Oe‑FKBP4‑EGFP and Ad‑siRNA‑FKBP4‑EGFP were constructed to examine the association among the above factors using the RT‑qPCR and WB methods. In the animal experiment, the vaginal smear, H&E staining and ELISA results showed that the PCOS model was successfully established. The IHC staining revealed that the expression of FKBP52 in the GCs of the PCOS model group was higher than the remaining groups (P<0.01). The mRNA and expression levels of FKBP52 and AR in the PCOS model rats were significantly increased, when compared with levels in the other rats (P<0.05). The expression level of p‑ERK1/2 was also higher (P<0.05). In the GC experiment, following overexpression of the FKBP4 gene, the mRNA and expression levels of FKBP52 and AR were increased (P<0.05). The expression level of p‑ERK1/2 was also increased (P<0.05). Following FKBP4 gene silencing, the mRNA and expression levels of FKBP52 and AR were decreased (P<0.05). The expression level of ERK1/2 was also decreased (P<0.05). However, the expression level of p‑ERK1/2 was increased (P<0.05). In conclusion, the upregulation of co‑chaperone FKBP52 may mediate the activation of AR through the MAPK/ERK pathway.
Figures




References
-
- Asemi Z, Samimi M, Taghizadeh M, Esmaillzadeh A. Effects of Ramadan fasting on glucose homeostasis, lipid profiles, inflammation and oxidative stress in women with polycystic ovary syndrome in Kashan, Iran. Arch Iran Med. 2015;18:806–810. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

