Effects of VEGFR1+ hematopoietic progenitor cells on pre-metastatic niche formation and in vivo metastasis of breast cancer cells
- PMID: 30483898
- PMCID: PMC6373264
- DOI: 10.1007/s00432-018-2802-6
Effects of VEGFR1+ hematopoietic progenitor cells on pre-metastatic niche formation and in vivo metastasis of breast cancer cells
Abstract
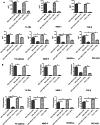
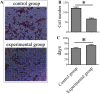
The pre-metastatic niche has been shown to play a critical role in tumor metastasis, and its formation is closely related to the tumor microenvironment. However, the underlying molecular mechanisms remain unclear. In the present study, we successfully established a mouse model of lung metastasis using luciferase-expressing MDA-MB-435s cells. In this model, recruitment of vascular endothelial growth factor receptor-1 (VEGFR1)+CD133+ hematopoietic progenitor cells (HPCs) was gradually increased in lung but gradually decreased after the formation of tumor colonies in lung. We also established a highly metastatic MDA-MB-435s (MDA-MB-435s-HM) cell line from the mouse model. Changes in protein profiles in different culture conditions were investigated by protein microarray analysis. The levels of CXC chemokine ligand 16, interleukin (IL)-2Rα, IL-2Rγ, matrix metalloproteinase (MMP)-1, MMP-9, platelet-derived growth factor receptor (PDGFR)-α, stromal cell-derived factor (SDF)-1α, transforming growth factor (TGF)-β, platelet endothelial cell adhesion molecule (PECAM)-1 and vascular endothelial (VE)-cadherin were significantly greater (> fivefold) in the culture medium from MDA-MB-435s-HM cells than in that from MDA-MB-435s cells. Moreover, the levels of MMP-9, PDGFR-α, and PECAM-1 were significantly greater in the co-culture medium of MDA-MB-435s-HM cells and CD133+ HPCs than in that from MDA-MB-435s-HM cells. Differentially expressed proteins were validated by enzyme-linked immunosorbent assay, and expression of their transcripts was confirmed by quantitative real-time polymerase chain reaction. Moreover, inhibition of MMP-9, PDGFR-α, and PECAM-1 by their specific inhibitors or antibodies significantly decreased cell migration, delayed lung metastasis, and decreased recruitment of VEGFR1+CD133+ HPCs into lung. Intra-hepatic growth of HPCs enhanced the invasive growth of MDA-MB-435s-HM cells in the liver. Our data indicate that VEGFR1+CD133+ HPCs contribute to lung metastasis.
Keywords: Hematopoietic progenitor cells; Lung metastasis; Pre-metastatic niche; Protein microarray; VEGFR1.
Conflict of interest statement
All the authors declare that there is no conflict of interest in this work.
Figures




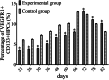


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

