Recent Advances in X-Ray Hydroxyl Radical Footprinting at the Advanced Light Source Synchrotron
- PMID: 30484401
- PMCID: PMC7655235
- DOI: 10.2174/0929866526666181128125725
Recent Advances in X-Ray Hydroxyl Radical Footprinting at the Advanced Light Source Synchrotron
Abstract
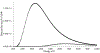
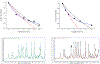
Background: Synchrotron hydroxyl radical footprinting is a relatively new structural method used to investigate structural features and conformational changes of nucleic acids and proteins in the solution state. It was originally developed at the National Synchrotron Light Source at Brookhaven National Laboratory in the late nineties, and more recently, has been established at the Advanced Light Source at Lawrence Berkeley National Laboratory. The instrumentation for this method is an active area of development, and includes methods to increase dose to the samples while implementing high-throughput sample delivery methods.
Conclusion: Improving instrumentation to irradiate biological samples in real time using a sample droplet generator and inline fluorescence monitoring to rapidly determine dose response curves for samples will significantly increase the range of biological problems that can be investigated using synchrotron hydroxyl radical footprinting.
Keywords: Advanced Light Source; Synchrotron; beamline; footprinting; hydroxyl radical footprinting; radiolysis; radiolytic labeling; x-ray..
Copyright© Bentham Science Publishers; For any queries, please email at epub@benthamscience.net.
Conflict of interest statement
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
Figures

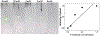

Similar articles
-
Development of Container Free Sample Exposure for Synchrotron X-ray Footprinting.Anal Chem. 2020 Jan 7;92(1):1565-1573. doi: 10.1021/acs.analchem.9b04849. Epub 2019 Dec 16. Anal Chem. 2020. PMID: 31790200 Free PMC article.
-
New high-throughput endstation to accelerate the experimental optimization pipeline for synchrotron X-ray footprinting.J Synchrotron Radiat. 2021 Sep 1;28(Pt 5):1321-1332. doi: 10.1107/S1600577521005026. Epub 2021 Jul 20. J Synchrotron Radiat. 2021. PMID: 34475281 Free PMC article.
-
The Beamline X28C of the Center for Synchrotron Biosciences: a national resource for biomolecular structure and dynamics experiments using synchrotron footprinting.J Synchrotron Radiat. 2007 May;14(Pt 3):233-43. doi: 10.1107/S0909049507013118. Epub 2007 Apr 11. J Synchrotron Radiat. 2007. PMID: 17435298
-
Development of Synchrotron Footprinting at NSLS and NSLS-II.Protein Pept Lett. 2019;26(1):55-60. doi: 10.2174/0929866526666181128125125. Protein Pept Lett. 2019. PMID: 30484397 Review.
-
Recent Advances and Applications in Synchrotron X-Ray Protein Footprinting for Protein Structure and Dynamics Elucidation.Protein Pept Lett. 2016;23(3):309-22. doi: 10.2174/0929866523666160201150057. Protein Pept Lett. 2016. PMID: 26833224 Review.
Cited by
-
An automated liquid jet for fluorescence dosimetry and microsecond radiolytic labeling of proteins.Commun Biol. 2022 Aug 25;5(1):866. doi: 10.1038/s42003-022-03775-1. Commun Biol. 2022. PMID: 36008591 Free PMC article.
-
Development of Container Free Sample Exposure for Synchrotron X-ray Footprinting.Anal Chem. 2020 Jan 7;92(1):1565-1573. doi: 10.1021/acs.analchem.9b04849. Epub 2019 Dec 16. Anal Chem. 2020. PMID: 31790200 Free PMC article.
-
Advances in mass spectrometry-based footprinting of membrane proteins.Proteomics. 2022 Apr;22(8):e2100222. doi: 10.1002/pmic.202100222. Proteomics. 2022. PMID: 35290716 Free PMC article. Review.
References
-
- Xu G; Chance MR Hydroxyl radical-mediated modification of proteins as probes for structural proteomics. Chem. Rev, 2007, 107(8), 3514–3543. - PubMed
-
- Liljenzin J Radiation Effects on Matter. In: Radiochemistry and Nuclear Chemistry, 3rd ed.; Choppin GR; ed.; Butterworth-Heinemann: Oxford, United Kingdom, 2002.
-
- Gupta S; Sullivan M; Toomey J; Kiselar J; Chance MR The beamline X28C of the center for synchrotron biosciences: A national resource for biomolecular structure and dynamics experiments using synchrotron footprinting. J. Synchrotron Radiat, 2007, 14(Pt 3), 233–243. - PubMed

