Nobiletin Enhances Chemosensitivity to Adriamycin through Modulation of the Akt/GSK3β/β⁻Catenin/MYCN/MRP1 Signaling Pathway in A549 Human Non-Small-Cell Lung Cancer Cells
- PMID: 30486290
- PMCID: PMC6316077
- DOI: 10.3390/nu10121829
Nobiletin Enhances Chemosensitivity to Adriamycin through Modulation of the Akt/GSK3β/β⁻Catenin/MYCN/MRP1 Signaling Pathway in A549 Human Non-Small-Cell Lung Cancer Cells
Abstract
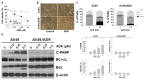
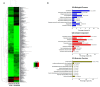
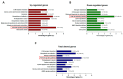
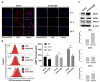
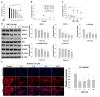
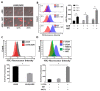
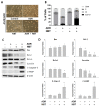
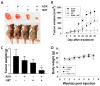
Drug resistance is a major problem in the treatment of non-small-cell lung cancer (NSCLC). In this study, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis was performed to identify the differentially expressed genes in Adriamycin (ADR)-resistant NSCLC A549/ADR cells compared with parental A549 cells. Among the tested phytochemicals, nobiletin (NBT) is able to overcome the ADR resistance of A549/ADR cells. NBT treatment decreased the expression of a neuroblastoma-derived MYC (MYCN) and multidrug resistance-associated protein 1 (MRP1) as well as downregulating Akt, GSK3β, and β-catenin. Consistent with these results, NBT treatment resulted in the accumulation of intracellular ADR. A combination index (CI) assay confirmed the synergistic effect of combined treatment with NBT and ADR in reducing the viability of A549/ADR cells (CI = 0.152). Combined treatment with NBT and ADR enhanced apoptosis in A549/ADR cells, as evidenced by increased caspase-3 activation, poly (ADP-ribose) polymerase (PARP) cleavage, and sub-G1 population compared to treatment with ADR alone. In vivo experiments using a mouse xenograft model revealed that combination therapy with NBT and ADR significantly reduced tumor volume by 84.15%. These data suggest that NBT can sensitize ADR-induced cytotoxicity against A549/ADR cells by inhibiting MRP1 expression, indicating that NBT could serve as an effective adjuvant agent for ADR-based chemotherapy in lung cancer.
Keywords: A549 human non-small-cell lung cancer cells; Adriamycin (ADR); multidrug resistance-associated protein 1 (MRP1); nobiletin (NBT).
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Ferlay J., Soerjomataram I., Ervik M., Dikshit R., Eser S., Mathers C., Rebelo M., Parkin D., Forman D., Bray F. Cancer Incidence and Mortality Worldwide: Iarc Cancer Base no. 11. International Agency for Research on Cancer; Paris, France: 2013.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

