Hypoxia potentiates gemcitabine-induced stemness in pancreatic cancer cells through AKT/Notch1 signaling
- PMID: 30486896
- PMCID: PMC6263055
- DOI: 10.1186/s13046-018-0972-3
Hypoxia potentiates gemcitabine-induced stemness in pancreatic cancer cells through AKT/Notch1 signaling
Abstract
Background: Profound chemoresistance remains an intractable obstacle in pancreatic cancer treatment. Pancreatic cancer stem cells (CSCs) and the ubiquitous hypoxic niche have been proposed to account for drug resistance. However, the mechanism involved requires further exploration. This study investigated whether the hypoxic niche enhances gemcitabine-induced stemness and acquired resistance in pancreatic cancer cells by activating the AKT/Notch1 signaling cascade. The therapeutic effects of blockading this signaling cascade on gemcitabine-enriched CSCs were also investigated.
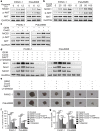
Methods: The expression levels of CSC-associated markers Bmi1 and Sox2 as well as those of proteins involved in AKT/Notch1 signaling were measured by Western blot analysis. The expression level of the pancreatic CSC marker CD24 was measured by flow cytometry. Change in gemcitabine sensitivity was evaluated by the MTT assay. The ability of sphere formation was tested by the sphere-forming assay in stem cell medium. The ability of migration and invasion was detected by the transwell migration/invasion assay. A mouse xenograft model of pancreatic cancer was established to determine the effect of Notch1 inhibition on the killing effect of gemcitabine in vivo. The ability of metastasis was investigated by an in vivo lung metastasis assay.
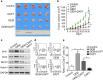
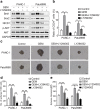
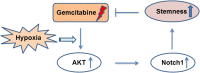
Results: Gemcitabine promoted pancreatic cancer cell stemness and associated malignant phenotypes such as enhanced migration, invasion, metastasis, and chemoresistance. The AKT/Notch1 signaling cascade was activated after gemcitabine treatment and mediated this process. Blockading this pathway enhanced the killing effect of gemcitabine in vivo. However, supplementation with hypoxia treatment synergistically enhanced the AKT/Notch1 signaling pathway and collaboratively promoted gemcitabine-induced stemness.
Conclusions: These findings demonstrate a novel mechanism of acquired gemcitabine resistance in pancreatic cancer cells through induction of stemness, which was mediated by the activation of AKT/Notch1 signaling and synergistically aggravated by the ubiquitous hypoxic niche. Our results might provide new insights for identifying potential targets for reversing chemoresistance in patients with pancreatic cancer.
Keywords: AKT; Cancer stem cell; Gemcitabine; Hypoxia; Notch1.
Conflict of interest statement
Ethics approval and consent to participate
This study was approved by the ethical review board of Renmin Hospital, Wuhan University (Wuhan, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures



References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

