MopA, the Mn Oxidizing Protein From Erythrobacter sp. SD-21, Requires Heme and NAD+ for Mn(II) Oxidation
- PMID: 30487779
- PMCID: PMC6247904
- DOI: 10.3389/fmicb.2018.02671
MopA, the Mn Oxidizing Protein From Erythrobacter sp. SD-21, Requires Heme and NAD+ for Mn(II) Oxidation
Abstract
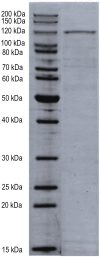
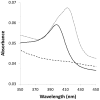
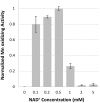
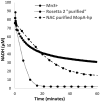
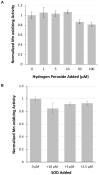
Bacterial manganese (Mn) oxidation is catalyzed by a diverse group of microbes and can affect the fate of other elements in the environment. Yet, we understand little about the enzymes that catalyze this reaction. The Mn oxidizing protein MopA, from Erythrobacter sp. strain SD-21, is a heme peroxidase capable of Mn(II) oxidation. Unlike Mn oxidizing multicopper oxidase enzymes, an understanding of MopA is very limited. Sequence analysis indicates that MopA contains an N-terminal heme peroxidase domain and a C-terminal calcium binding domain. Heterologous expression and nickel affinity chromatography purification of the N-terminal peroxidase domain (MopA-hp) from Erythrobacter sp. strain SD-21 led to partial purification. MopA-hp is a heme binding protein that requires heme, NAD+, and calcium (Ca2+) for activity. Mn oxidation is also stimulated by the presence of pyrroloquinoline quinone. MopA-hp has a K M for Mn(II) of 154 ± 46 μM and k cat = 1.6 min-1. Although oxygen requiring MopA-hp is homologous to peroxidases based on sequence, addition of hydrogen peroxide and hydrogen peroxide scavengers had little effect on Mn oxidation, suggesting this is not the oxidizing agent. These studies provide insight into the mechanism by which MopA oxidizes Mn.
Keywords: Mn; MopA; NAD+; PQQ; heme; lactoperoxidase; manganese; peroxidase cyclooxygenase.
Figures
Similar articles
-
Heterologous expression and characterization of the manganese-oxidizing protein from Erythrobacter sp. strain SD21.Appl Environ Microbiol. 2014 Nov;80(21):6837-42. doi: 10.1128/AEM.01873-14. Epub 2014 Aug 29. Appl Environ Microbiol. 2014. PMID: 25172859 Free PMC article.
-
Mn(II) oxidation is catalyzed by heme peroxidases in "Aurantimonas manganoxydans" strain SI85-9A1 and Erythrobacter sp. strain SD-21.Appl Environ Microbiol. 2009 Jun;75(12):4130-8. doi: 10.1128/AEM.02890-08. Epub 2009 May 1. Appl Environ Microbiol. 2009. PMID: 19411418 Free PMC article.
-
Identification of a Third Mn(II) Oxidase Enzyme in Pseudomonas putida GB-1.Appl Environ Microbiol. 2016 Jun 13;82(13):3774-3782. doi: 10.1128/AEM.00046-16. Print 2016 Jul 1. Appl Environ Microbiol. 2016. PMID: 27084014 Free PMC article.
-
Bacteriogenic manganese oxides.Acc Chem Res. 2010 Jan 19;43(1):2-9. doi: 10.1021/ar800232a. Acc Chem Res. 2010. PMID: 19778036 Review.
-
Geomicrobiology of manganese(II) oxidation.Trends Microbiol. 2005 Sep;13(9):421-8. doi: 10.1016/j.tim.2005.07.009. Trends Microbiol. 2005. PMID: 16054815 Review.
Cited by
-
A soil-borne Mn(II)-oxidizing bacterium of Providencia sp. exploits a strategy of superoxide production coupled to hydrogen peroxide consumption to generate Mn oxides.Arch Microbiol. 2022 Feb 12;204(3):168. doi: 10.1007/s00203-022-02771-7. Arch Microbiol. 2022. PMID: 35152320
-
Catalase-peroxidase StKatG2 from Salinicola tamaricis: a versatile Mn(II) oxidase that decolorizes malachite green.Front Microbiol. 2024 Nov 5;15:1478305. doi: 10.3389/fmicb.2024.1478305. eCollection 2024. Front Microbiol. 2024. PMID: 39564493 Free PMC article.
-
Substrate Specificity of Biofilms Proximate to Historic Shipwrecks.Microorganisms. 2023 Sep 27;11(10):2416. doi: 10.3390/microorganisms11102416. Microorganisms. 2023. PMID: 37894074 Free PMC article.
-
Manganese-contaminated groundwater treatment by novel bacterial isolates: kinetic study and mechanism analysis using synchrotron-based techniques.Sci Rep. 2020 Aug 7;10(1):13391. doi: 10.1038/s41598-020-70355-w. Sci Rep. 2020. PMID: 32770016 Free PMC article.
-
Comparative Genomics on Cultivated and Uncultivated Freshwater and Marine "Candidatus Manganitrophaceae" Species Implies Their Worldwide Reach in Manganese Chemolithoautotrophy.mBio. 2022 Apr 26;13(2):e0342121. doi: 10.1128/mbio.03421-21. Epub 2022 Mar 14. mBio. 2022. PMID: 35285693 Free PMC article.
References
-
- Anderson C. R., Davis R. E., Bandolin N. S., Baptista A. M., Tebo B. M. (2011). Analysis of in situ manganese(II) oxidation in the Columbia River and offshore plume: linking Aurantimonas and the associated microbial community to an active biogeochemical cycle. Environ. Microbiol. 13 1561–1576. 10.1111/j.1462-2920.2011.02462.x - DOI - PMC - PubMed
-
- Anderson C. R., Johnson H. A., Caputo N., Davis R. E., Torpey J. W., Tebo B. M. (2009). Mn(II) oxidation is catalyzed by heme peroxidases in “Aurantimonas manganoxydans” strain SI85-9A1 and Erythrobacter sp. strain SD-21. Appl. Environ. Microbiol. 75 4130–4138. 10.1128/AEM.02890-08 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

