The Role of Autophagy in iNKT Cell Development
- PMID: 30487800
- PMCID: PMC6246678
- DOI: 10.3389/fimmu.2018.02653
The Role of Autophagy in iNKT Cell Development
Abstract
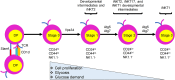
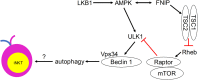
CD1d-restricted invariant natural killer T (iNKT) cells are innate-like T cells that express an invariant T cell receptor (TCR) α-chain and recognize self and foreign glycolipid antigens. They can rapidly respond to agonist activation and stimulate an extensive array of immune responses. Thymic development and function of iNKT cells are regulated by many different cellular processes, including autophagy, a self-degradation mechanism. In this mini review, we discuss the current understanding of how autophagy regulates iNKT cell development and effector lineage differentiation. Importantly, we propose that iNKT cell development is tightly controlled by metabolic reprogramming.
Keywords: CD1d; autophagy; invariant natural killer T cells; metabolic switch; thymic development.
Figures
Similar articles
-
Essential role for autophagy during invariant NKT cell development.Proc Natl Acad Sci U S A. 2014 Dec 30;111(52):E5678-87. doi: 10.1073/pnas.1413935112. Epub 2014 Dec 15. Proc Natl Acad Sci U S A. 2014. PMID: 25512546 Free PMC article.
-
New Genetically Manipulated Mice Provide Insights Into the Development and Physiological Functions of Invariant Natural Killer T Cells.Front Immunol. 2018 Jun 14;9:1294. doi: 10.3389/fimmu.2018.01294. eCollection 2018. Front Immunol. 2018. PMID: 29963043 Free PMC article. Review.
-
Critical role for invariant chain in CD1d-mediated selection and maturation of Vα14-invariant NKT cells.Immunol Lett. 2011 Sep 30;139(1-2):33-41. doi: 10.1016/j.imlet.2011.04.012. Epub 2011 May 5. Immunol Lett. 2011. PMID: 21565221 Free PMC article.
-
The ins and outs of type I iNKT cell development.Mol Immunol. 2019 Jan;105:116-130. doi: 10.1016/j.molimm.2018.09.023. Epub 2018 Nov 28. Mol Immunol. 2019. PMID: 30502719 Free PMC article. Review.
-
T Cell Receptor Expression Timing and Signal Strength in the Functional Differentiation of Invariant Natural Killer T Cells.Front Immunol. 2019 Apr 26;10:841. doi: 10.3389/fimmu.2019.00841. eCollection 2019. Front Immunol. 2019. PMID: 31080448 Free PMC article. Review.
Cited by
-
Cullin 3-Mediated Regulation of Intracellular Iron Homeostasis Promotes Thymic Invariant NKT Cell Maturation.Immunohorizons. 2023 Mar 1;7(3):235-242. doi: 10.4049/immunohorizons.2300002. Immunohorizons. 2023. PMID: 36951874 Free PMC article.
-
Unveiling potential drug targets for hyperparathyroidism through genetic insights via Mendelian randomization and colocalization analyses.Sci Rep. 2024 Mar 18;14(1):6435. doi: 10.1038/s41598-024-57100-3. Sci Rep. 2024. PMID: 38499600 Free PMC article.
-
Autophagy and Its Lineage-Specific Roles in the Hematopoietic System.Oxid Med Cell Longev. 2023 May 3;2023:8257217. doi: 10.1155/2023/8257217. eCollection 2023. Oxid Med Cell Longev. 2023. PMID: 37180758 Free PMC article. Review.
-
Roles and therapeutic potential of CD1d-Restricted NKT cells in inflammatory skin diseases.Front Immunol. 2022 Sep 2;13:979370. doi: 10.3389/fimmu.2022.979370. eCollection 2022. Front Immunol. 2022. PMID: 36119077 Free PMC article. Review.
-
Pik3c3 deficiency in myeloid cells imparts partial resistance to experimental autoimmune encephalomyelitis associated with reduced IL-1β production.Cell Mol Immunol. 2021 Aug;18(8):2024-2039. doi: 10.1038/s41423-020-00589-1. Epub 2020 Nov 24. Cell Mol Immunol. 2021. PMID: 33235386 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

