Aberrant DNA damage response and DNA repair pathway in high glucose conditions
- PMID: 30498558
- PMCID: PMC6258084
Aberrant DNA damage response and DNA repair pathway in high glucose conditions
Abstract
Background: Higher cancer rates and more aggressive behavior of certain cancers have been reported in populations with diabetes mellitus. This association has been attributed in part to the excessive reactive oxygen species generated in diabetic conditions and to the resulting oxidative DNA damage. It is not known, however, whether oxidative stress is the only contributing factor to genomic instability in patients with diabetes or whether high glucose directly also affects DNA damage and repair pathways.
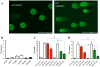
Results: Normal renal epithelial cells and renal cell carcinoma cells are more chemo- and radiation resistant when cultured in high concentrations of glucose. In high glucose conditions, the CHK1-mediated DNA damage response is not activated properly. Cells in high glucose also have slower DNA repair rates and accumulate more mutations than cells grown in normal glucose concentrations. Ultimately, these cells develop a transforming phenotype.
Conclusions: In high glucose conditions, defective DNA damage responses most likely contribute to the higher mutation rate in renal epithelial cells, in addition to oxidative DNA damage. The DNA damage and repair are normal enzyme dependent mechanisms requiring euglycemic environments. Aberrant DNA damage response and repair in cells grown in high glucose conditions underscore the importance of maintaining good glycemic control in patients with diabetes mellitus and cancer.
Keywords: ATR; Chemo resistant; DNA damage response; Diabetes; checkpoint kinase 1.
Conflict of interest statement
Competing Financial Interests None.
Figures
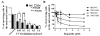



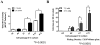
References
-
- Vigneri P, Frasca F, Sciacca L, Pandini G, Vigneri R. Diabetes and cancer. Endocr Relat Cancer. 2009;16:1103–23. - PubMed
-
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–25. - PubMed
-
- Kastan MB, Bartek J. Cell-cycle checkpoints and cancer. Nature. 2004;432:316–323. - PubMed
-
- Zhou BB, Elledge SJ. The DNA damage response: putting checkpoints in perspective. Nature. 2000;408:433–9. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
