TPT sulfonate, a single, oral dose schistosomicidal prodrug: In vivo efficacy, disposition and metabolic profiling
- PMID: 30503203
- PMCID: PMC6287543
- DOI: 10.1016/j.ijpddr.2018.10.004
TPT sulfonate, a single, oral dose schistosomicidal prodrug: In vivo efficacy, disposition and metabolic profiling
Abstract
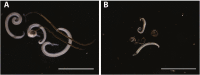
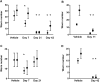
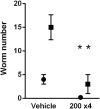
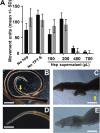
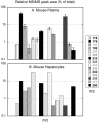
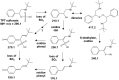
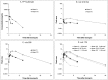
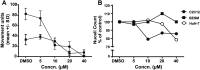
Treatment of schistosomiasis relies precariously on just one drug, praziquantel (PZQ). In the search for alternatives, 15 S-[2-(alkylamino)alkane] thiosulfuric acids were obtained from a previous research program and profiled in mice for efficacy against both mature (>42-day-old) and juvenile (21-day-old) Schistosoma mansoni using a screening dose of 100 mg/kg PO QDx4. One compound, S-[2-(tert-butylamino)-1-phenylethane] thiosulfuric acid (TPT sulfonate), was the most effective by decreasing female and male worm burdens by ≥ 90% and ≥46% (mature), and ≥89% and ≥79% (juvenile), respectively. In contrast, PZQ decreased mature female and male worm burdens by 95% and 94%, respectively, but was ineffective against juvenile stages. Against 7-day-old lung-stage worms, TPT sulfonate was only effective at twice the dose decreasing female and male burdens by 95 and 80%, respectively. Single oral doses at 400 and/or 600 mg/kg across various developmental time-points (1-, 7-, 15-, 21- and/or 42 day-old) were consistent with the QD x4 data; efficacy was strongest once the parasites had completed lung migration, and female and male burdens were decreased by at least 90% and 80%, respectively. In vitro, TPT sulfonate is inactive against the parasite suggesting a pro-drug mechanism of action. In mice, TPT sulfonate is fully absorbed and subject to rapid, non-CYP-mediated, first-pass metabolism that is initiated by desulfation and yields a series of metabolites. The initially-formed free thiol-containing metabolite, termed TP thiol, was chemically synthesized; it dose-dependently decreased S. mansoni and Schistosoma haematobium motility in vitro. Also, when administered as a single 50 mg/kg IP dose, TP thiol decreased 33-day-old S. mansoni female and male burdens by 35% and 44%, with less severe organomegaly. Overall, TPT sulfonate's efficacy profile is competitive with that of PZQ. Also, the characterization of a parasiticidal metabolite facilitates an understanding and improvement of the chemistry, and identification of the mechanism of action and/or target.
Keywords: Alkylaminoalkanethiosulfuric acid; Anthelmintic; Drug disposition; Drug metabolism; Schistosoma; Schistosomiasis.
Copyright © 2018 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures




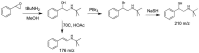
References
-
- Abdulla M.H., Ruelas D.S., Wolff B., Snedecor J., Lim K.C., Xu F., Renslo A.R., Williams J., McKerrow J.H., Caffrey C.R. Drug discovery for schistosomiasis: hit and lead compounds identified in a library of known drugs by medium-throughput phenotypic screening. PLoS Neglected Trop. Dis. 2009;3:e478. - PMC - PubMed
-
- Andrews P. Praziquantel: mechanisms of anti-schistosomal activity. Pharmacol. Ther. 1985;29:129–156. - PubMed
-
- Andrews P., Thomas H., Pohlke R., Seubert J. Praziquantel. Med Res Rev. 1983;3:147–200. - PubMed
-
- Basch P.F. Cultivation of Schistosoma mansoni in vitro. I. Establishment of cultures from cercariae and development until pairing. J. Parasitol. 1981;67:179–185. - PubMed

