Recent developments in topoisomerase-targeted cancer chemotherapy
- PMID: 30505655
- PMCID: PMC6251812
- DOI: 10.1016/j.apsb.2018.07.008
Recent developments in topoisomerase-targeted cancer chemotherapy
Abstract
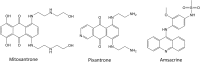
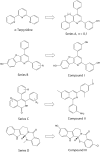
The DNA topoisomerase enzymes are essential to cell function and are found ubiquitously in all domains of life. The various topoisomerase enzymes perform a wide range of functions related to the maintenance of DNA topology during DNA replication, and transcription are the targets of a wide range of antimicrobial and cancer chemotherapeutic agents. Natural product-derived agents, such as the camptothecin, anthracycline, and podophyllotoxin drugs, have seen broad use in the treatment of many types of cancer. Selective targeting of the topoisomerase enzymes for cancer treatment continues to be a highly active area of basic and clinical research. The focus of this review will be to summarize the current state of the art with respect to clinically used topoisomerase inhibitors for targeted cancer treatment and to discuss the pharmacology and chemistry of promising new topoisomerase inhibitors in clinical and pre-clinical development.
Keywords: ADP, adenosine diphosphate; AQD, anti-cancer quinolone derivative; ATP, adenosine triphosphate; Cancer; Clinical; DNA, deoxyribonucleic acid; Inhibition; MTD, maximum tolerated dose; NCI, National Cancer Institute; NCT, national clinical trial; Oncology; PD, pharmacodynamics; PFS, progression free survival; Pre-clinical; Topoisomerase.
Figures









References
-
- Champoux J.J. DNA topoisomerases: structure, function, and mechanism. Annu Rev Biochem. 2001;70:369–413. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

