Enrich and Expand Rare Antigen-specific T Cells with Magnetic Nanoparticles
- PMID: 30507913
- PMCID: PMC6724533
- DOI: 10.3791/58640
Enrich and Expand Rare Antigen-specific T Cells with Magnetic Nanoparticles
Abstract
We have developed a tool to both enrich and expand antigen-specific T cells. This can be helpful in cases such as to A) detect the existence of antigen-specific T cells, B) probe the dynamics of antigen-specific responses, C) understand how antigen-specific responses affect disease state such as autoimmunity, D) demystify heterogeneous responses for antigen-specific T cells, or E) utilize antigen-specific cells for therapy. The tool is based on a magnetic particle that we conjugate antigen-specific and T cell co-stimulatory signals, and that we term as artificial antigen presenting cells (aAPCs). Consequently, since the technology is simple to produce, it can easily be adopted by other laboratories; thus, our purpose here is to describe in detail the fabrication and subsequent use of the aAPCs. We explain how to attach antigen-specific and co-stimulatory signals to the aAPCs, how to utilize them to enrich for antigen-specific T cells, and how to expand antigen-specific T cells. Furthermore, we will highlight engineering design considerations based on experimental and biological information of our experience with characterizing antigen-specific T cells.
Figures
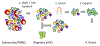

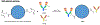

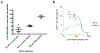
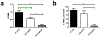



Similar articles
-
Biodegradable Cationic Polymer Blends for Fabrication of Enhanced Artificial Antigen Presenting Cells to Treat Melanoma.ACS Appl Mater Interfaces. 2021 Feb 24;13(7):7913-7923. doi: 10.1021/acsami.0c19955. Epub 2021 Feb 12. ACS Appl Mater Interfaces. 2021. PMID: 33573372 Free PMC article.
-
Development of third generation anti-EGFRvIII chimeric T cells and EGFRvIII-expressing artificial antigen presenting cells for adoptive cell therapy for glioma.PLoS One. 2018 Jul 5;13(7):e0199414. doi: 10.1371/journal.pone.0199414. eCollection 2018. PLoS One. 2018. PMID: 29975720 Free PMC article.
-
Efficient magnetic enrichment of antigen-specific T cells by engineering particle properties.Biomaterials. 2018 Dec;187:105-116. doi: 10.1016/j.biomaterials.2018.09.029. Epub 2018 Sep 28. Biomaterials. 2018. PMID: 30312851 Free PMC article.
-
Nanoscale artificial antigen presenting cells for cancer immunotherapy.Mol Immunol. 2018 Jun;98:13-18. doi: 10.1016/j.molimm.2018.02.016. Epub 2018 Mar 7. Mol Immunol. 2018. PMID: 29525074 Free PMC article. Review.
-
Particle-Based Artificial Antigen-Presenting Cell Systems for T Cell Activation in Adoptive T Cell Therapy.ACS Nano. 2024 Mar 26;18(12):8571-8599. doi: 10.1021/acsnano.3c10180. Epub 2024 Mar 14. ACS Nano. 2024. PMID: 38483840 Review.
Cited by
-
Ex vivo expansion and hydrogel-mediated in vivo delivery of tissue-resident memory T cells for immunotherapy.Sci Adv. 2024 Dec 13;10(50):eadm7928. doi: 10.1126/sciadv.adm7928. Epub 2024 Dec 13. Sci Adv. 2024. PMID: 39671478 Free PMC article.
-
In Vivo Stimulation of Therapeutic Antigen-Specific T Cells in an Artificial Lymph Node Matrix.Adv Mater. 2024 Jun;36(23):e2310043. doi: 10.1002/adma.202310043. Epub 2024 Mar 1. Adv Mater. 2024. PMID: 38358310 Free PMC article.
References
-
- Prasad V immunotherapy: Tisagenlecleucel-the first approved Car-t-cell therapy: implications for payers and policy makers. Nature Reviews Clinical Oncology. 15 (1), 11 (2018). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
