Confirmation of HCV viremia using HCV RNA and core antigen testing on dried blood spot in HIV infected peoples who inject drugs in Vietnam
- PMID: 30514229
- PMCID: PMC6280470
- DOI: 10.1186/s12879-018-3529-3
Confirmation of HCV viremia using HCV RNA and core antigen testing on dried blood spot in HIV infected peoples who inject drugs in Vietnam
Abstract
Background: Nucleic acid tests performed on blood samples collected on Dried Blood Spot (DBS) and detection of HCV core antigen (HCVcAg) are two approaches that may facilitate access to HCV diagnosis in low and middle incomes countries. In this study we evaluate HCV RNA and HCV antigen testing on DBS in HIV/HCV co-infected peoples who inject drugs in Vietnam.
Method: One hundred and four HIV/HCV seropositive patients managed in outpatient care at the Haiphong Viet Tiep hospital were included in this study from February to March, 2014 (ANRS 12262 study).
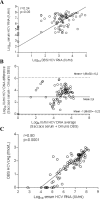
Results: Eighty-six subjects were tested positive for HCV RNA in serum, median (IQR): 6.9 log10 IU/ml (5.6-7.4 log10 IU/ml). Genotypes consisted of 57 G1 (69%), 3 G3 (4%), and 22 G6 (27%). HCV RNA was detected on DBS specimens in 79 out 86 subjects with chronic hepatitis C (sensitivity 92.5%; 95% CI: 85.1-96.9%). HCV RNA level on DBS and serum was moderately correlated (r = 0.24; p = 0.05) suggesting a degradation of HCV RNA due to transportation and storage conditions. HCVcAg was detected in 75/86 dB specimens (sensitivity: 87.2%; 95% CI: 78.3-93.4%), with a strong positive relationship between DBS HCVcAg and serum HCV RNA levels (r = 0.80; P < 0.0001).
Conclusions: Quantification of HCVcAg on DBS appears to benefit from substantial stability under prolonged storage conditions but with a lower analytical sensitivity compared to DBS HCV RNA testing. Detection of HCV RNA on DBS is an interesting approach for confirming viral replication in HCV seropositive persons but the impact of pre-analytical conditions on the integrity of HCV RNA needs to be controlled.
Keywords: HCV RNA; HCV core antigen; Hepatitis C virus (HCV); Human immunodeficiency virus (HIV); Vietnam.
Conflict of interest statement
Ethics approval and consent to participate
The study protocol and written informed consent form were approved by the Ethic Committee of the Viet Tiep Hospital and the Institutional Review Board (IRB) of the Haiphong Medical Services (n° 01BVVT/HDKH). Then, the written informed consent was obtained from all participants before blood sampling. Privacy and confidentiality as well as the anonymity of the participants were ensured at all stages of data collection.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures

References
-
- WHO . Combating hepatitis B and C to reach elimination by 2030. 2016.
-
- WHO. Global health sector strategy on viral hepatitis, 2016–2021. Available from: http://www.who.int/hepatitis/strategy2016-2021..
-
- WHO. Guidelines on Heaptitis B and C Testing 2017. Available from: http://apps.who.int/iris/bitstream/10665/254621/1/9789241549981-eng. Accessed 6 Feb 2017.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

