Coupling factors involved in preserving bone balance
- PMID: 30515522
- PMCID: PMC11105749
- DOI: 10.1007/s00018-018-2981-y
Coupling factors involved in preserving bone balance
Abstract
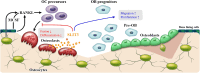
Coupling during bone remodeling refers to the spatial and temporal coordination of bone resorption with bone formation. Studies have assessed the subtle interactions between osteoclasts and osteoblasts to preserve bone balance. Traditionally, coupling research related to osteoclast function has focused on bone resorption activity causing the release of growth factors embedded in the bone matrix. However, considerable evidence from in vitro, animal, and human studies indicates the importance of the osteoclasts themselves in coupling phenomena, and many osteoclast-derived coupling factors have been identified. These include sphingosine-1-phosphate, vesicular-receptor activator of nuclear factor-κB, collagen triple helix repeat containing 1, and cardiotrophin-1. Interestingly, neuronal guidance molecules, such as slit guidance ligand 3, semaphorin (SEMA) 3A, SEMA4D, and netrin-1, originally identified as instructive cues allowing the navigation of growing axons to their targets, have been shown to be involved in the intercellular cross-talk among bone cells. This review discusses osteoclast-osteoblast coupling signals, including recent advances and the potential roles of these signals as therapeutic targets for osteoporosis and as biomarkers predicting human bone health.
Keywords: Axon guidance molecule; Biomarker; Bone remodeling; Coupling factor; Therapeutic target.
Conflict of interest statement
The authors declare that they have patents on S1P as a biomarker and SLIT3 as a therapeutic target for osteoporosis.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

