CTL019 (tisagenlecleucel): CAR-T therapy for relapsed and refractory B-cell acute lymphoblastic leukemia
- PMID: 30518999
- PMCID: PMC6237143
- DOI: 10.2147/DDDT.S138765
CTL019 (tisagenlecleucel): CAR-T therapy for relapsed and refractory B-cell acute lymphoblastic leukemia
Abstract
Over the past decades, survival of patients with acute lymphoblastic leukemia (ALL) has dramatically improved, but the subgroup of patients with relapsed/refractory ALL still continues to have dismal prognosis. As an emerging therapeutic approach, chimeric antigen receptor-modified T-cells (CAR-T) represent one of the few practice-changing therapies for this subgroup of patients. Originally conceived and built in Philadelphia (University of Pennsylvania), CTL019 or tisagenlecleucel, the first CAR-T approved by the US Food and Drug Administration, showed impressive results in refractory/relapsed ALL since the publication on two pediatric patients in 2013. It is in this context that we provide a review of this product in terms of manufacturing, pharmacology, toxicity, and efficacy studies. Evaluation and management of toxicities, particularly cytokine release syndrome and neurotoxicity, is recognized as an essential part of the patient treatment with broader use of IL-6 receptor inhibitor. An under-assessed aspect, the quality of life of patients entering CAR-T cells treatment, will also be reviewed. By their unique nature, CAR-T cells such as tisagenlecleucel operate in a different way than typical drugs, but also provide unique hope for B-cell malignancies.
Keywords: B-cell acute lymphoblastic leukemia; CTL019; tisagenlecleucel.
Conflict of interest statement
Disclosure Henrique Bittencourt and Pierre Teira are consulting for Novartis Oncology. The other authors report no conflicts of interest in this work.
Figures
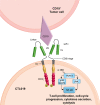
References
-
- Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review 1975–2014. Bethesda, MD: National Cancer Institute; 2017. [Accessed May 2018]. based on November 2016 SEER data submission. Available from: https://seer.cancer.gov/csr/1975_2014/
-
- Wolach O, Amitai I, Deangelo DJ. Current challenges and opportunities in treating adult patients with Philadelphia-negative acute lymphoblastic leukaemia. Br J Haematol. 2017;179(5):705–723. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

