Cryptococcus neoformans and Cryptococcus gattii clinical isolates from Thailand display diverse phenotypic interactions with macrophages
- PMID: 30520685
- PMCID: PMC6298761
- DOI: 10.1080/21505594.2018.1556150
Cryptococcus neoformans and Cryptococcus gattii clinical isolates from Thailand display diverse phenotypic interactions with macrophages
Abstract
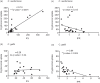
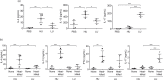
Cryptococcus-macrophage interaction is crucial in the development of cryptococcocal diseases. C. neoformans and C. gattii are major pathogenic species that occupy different niches and cause different clinical manifestations. However, the differences of macrophage interaction among these species in affecting different disease outcomes and immune responses have not been clearly addressed. Here, we examined the macrophage uptake rates, intracellular loads and intracellular proliferation rates of C. neoformans and C. gattii clinical isolates from Thailand and analyzed the effect of those interactions on fungal burdens and host immune responses. C. neoformans isolates showed a higher phagocytosis rate but lower intracellular proliferation rate than C. gattii. Indeed, the high intracellular proliferation rate of C. gattii isolates did not influence the fungal burdens in lungs and brains of infected mice, whereas infection with high-uptake C. neoformans isolates resulted in significantly higher brain burdens that associated with reduced survival rate. Interestingly, alveolar macrophages of mice infected with high-uptake C. neoformans isolates showed distinct patterns of alternatively activated macrophage (M2) gene expressions with higher Arg1, Fizz1, Il13 and lower Nos2, Ifng, Il6, Tnfa, Mcp1, csf2 and Ip10 transcripts. Corresponding to this finding, infection with high-uptake C. neoformans resulted in enhanced arginase enzyme activity, elevated IL-4 and IL-13 and lowered IL-17 in the bronchoalveolar lavage. Thus, our data suggest that the macrophage interaction with C. neoformans and C. gattii may affect different disease outcomes and the high phagocytosis rates of C. neoformans influence the induction of type-2 immune responses that support fungal dissemination and disease progression. Abbreviation: Arg1: Arginase 1; BAL: Bronchoalveolar lavage; CCL17: Chemokine (C-C motif) ligand 17; CNS: Central nervous system; CSF: Cerebrospinal fluid; Csf2: Colony-stimulating factor 2; Fizz1: Found in inflammatory zone 1; HIV: Human immunodeficiency virus; ICL: Intracellular cryptococcal load; Ifng: Interferon gamma; Ip10: IFN-g-inducible protein 10; IPR: Intracellular proliferation rate; Mcp1: Monocyte chemoattractant protein 1; Nos2: Nitric oxide synthase 2; PBS: Phosphate-Buffered Saline; Th: T helper cell; Tnfa: Tumor necrosis factor alpha.
Keywords: Cryptococcus gattii; Cryptococcus neoformans; fungal uptake; immune responses; macrophages.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
