Role of microparticles derived from monocytes, endothelial cells and platelets in the exacerbation of COPD
- PMID: 30532530
- PMCID: PMC6241682
- DOI: 10.2147/COPD.S175607
Role of microparticles derived from monocytes, endothelial cells and platelets in the exacerbation of COPD
Abstract
Background: Microparticles (MPs) are shedding membrane vesicles released from activated blood and endothelial cells under inflammatory conditions. The role of endothelial MPs (EMPs) in pathophysiology of COPD is relatively well known. However, the release and function of MPs of other cellular origins, eg, platelets, red blood cells and leukocytes, are not clearly evaluated in COPD.
Purpose: The aim of this study was to measure EMPs and other cell-derived circulating MPs in stable and exacerbated COPD patients.
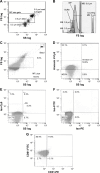
Patients and methods: A total of 50 patients with COPD and 19 healthy volunteers were enrolled in the study. EMPs (CD31+, CD62E+) and platelet-derived (CD61+, CD41+, CD42a+, PAC1+), red blood cell-derived (GlyA+) and leukocyte-derived (CD45+, CD13+, CD14+, CD56+) MPs were measured. Flow cytometry (FC) was performed on Beckman Coulter FC500 analyzer. MP reference gate was set using 0.3-0.5-0.9 µm microbeads with MP size gates of 0.5-1.0 µm.
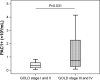
Results: All the measured MPs were significantly (P<0.001) higher in COPD patients than in the controls. Furthermore, CD62E+, CD41+, CD42a+ and CD14+ MP values were significantly (P<0.001) increased in exacerbated COPD compared to stable COPD. These MPs showed significant (P<0.001) inverse correlation with FEV1/FVC, as well.
Conclusion: In this study, we describe a reliable flow cytometric assay for MP analysis that was successfully applied in COPD. Besides EMPs, COPD is accompanied by an increased concentration of various MPs in the systemic circulation; particularly, platelet- and monocyte-derived MPs seem to be important in exacerbation.
Keywords: COPD; biomarker; cell-derived microparticles; flow cytometry.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures
References
-
- Hugel B, Martínez MC, Kunzelmann C, Freyssinet JM. Membrane microparticles: two sides of the coin. Physiology. 2005;20:22–27. - PubMed
-
- Vanwijk MJ, Vanbavel E, Sturk A, Nieuwland R. Microparticles in cardiovascular diseases. Cardiovasc Res. 2003;59(2):277–287. - PubMed
-
- Cocucci E, Racchetti G, Meldolesi J. Shedding microvesicles: artefacts no more. Trends Cell Biol. 2009;19(2):43–51. - PubMed
-
- Piccin A, Murphy WG, Smith OP. Circulating microparticles: pathophysiology and clinical implications. Blood Rev. 2007;21(3):157–171. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

