O-Fucosylation of thrombospondin-like repeats is required for processing of microneme protein 2 and for efficient host cell invasion by Toxoplasma gondii tachyzoites
- PMID: 30538131
- PMCID: PMC6369279
- DOI: 10.1074/jbc.RA118.005179
O-Fucosylation of thrombospondin-like repeats is required for processing of microneme protein 2 and for efficient host cell invasion by Toxoplasma gondii tachyzoites
Abstract
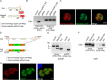
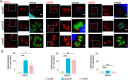
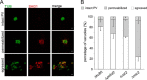
Toxoplasma gondii is an intracellular parasite that causes disseminated infections that can produce neurological damage in fetuses and immunocompromised individuals. Microneme protein 2 (MIC2), a member of the thrombospondin-related anonymous protein (TRAP) family, is a secreted protein important for T. gondii motility, host cell attachment, invasion, and egress. MIC2 contains six thrombospondin type I repeats (TSRs) that are modified by C-mannose and O-fucose in Plasmodium spp. and mammals. Here, using MS analysis, we found that the four TSRs in T. gondii MIC2 with protein O-fucosyltransferase 2 (POFUT2) acceptor sites are modified by a dHexHex disaccharide, whereas Trp residues within three TSRs are also modified with C-mannose. Disruption of genes encoding either POFUT2 or the putative GDP-fucose transporter (NST2) resulted in loss of MIC2 O-fucosylation, as detected by an antibody against the GlcFuc disaccharide, and in markedly reduced cellular levels of MIC2. Furthermore, in 10-15% of the Δpofut2 or Δnst2 vacuoles, MIC2 accumulated earlier in the secretory pathway rather than localizing to micronemes. Dissemination of tachyzoites in human foreskin fibroblasts was reduced for these knockouts, which both exhibited defects in attachment to and invasion of host cells comparable with the Δmic2 phenotype. These results, indicating that O-fucosylation of TSRs is required for efficient processing of MIC2 and for normal parasite invasion, are consistent with the recent demonstration that Plasmodium falciparum Δpofut2 strain has decreased virulence and also support a conserved role for this glycosylation pathway in quality control of TSR-containing proteins in eukaryotes.
Keywords: MIC2; O-fucosylation; Toxoplasma gondii; apicomplexa parasite; fucosyltransferase; glycoprotein secretion; glycosylation; invasion; protein glycosylation; thrombospondin-like repeat.
© 2019 Bandini et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures







References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

