BMP9 overexpressing adipose-derived mesenchymal stem cells promote cartilage repair in osteoarthritis-affected knee joint via the Notch1/Jagged1 signaling pathway
- PMID: 30542413
- PMCID: PMC6257276
- DOI: 10.3892/etm.2018.6754
BMP9 overexpressing adipose-derived mesenchymal stem cells promote cartilage repair in osteoarthritis-affected knee joint via the Notch1/Jagged1 signaling pathway
Abstract
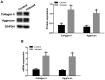
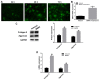
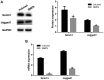
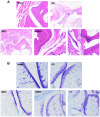
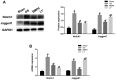
Osteoarthritis (OS) is a common disease in orthopedics. Although OS is known as an inflammation mediated by inflammatory cytokines; however, the mechanism is poorly understood. In the present study, the role of bone morphogenetic protein-9 (BMP9) was investigated in chondrogenic differentiation of adipose-derived mesenchymal stem cells (ADMSCs). ADMSCs were transfected with BMP9. BMP9 mRNA expression was detected by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). Type II collagen and aggrecan expression was detected by western blotting and RT-qPCR. Mouse models of knee OS were established. Hematoxylin-eosin staining and toluidine blue staining were performed to observe changes in the OS-affected knee joint. After intra-articular injection of ADMSCs transfected with BMP9, intra-articular expression of type II collagen and aggrecan was detected by western blot analysis and RT-qPCR. After the Notch signaling pathway was inhibited in ADMSCs, ADMSCs were injected into the articular cavity. The expression of Notch signaling pathway-related proteins Notch1 and Jagged1 was detected by western blot analysis and RT-qPCR. BMP9 promoted chondrogenic differentiation of ADMSCs. After injection of BMP9 overexpressing ADMSCs into the articular space, type II collagen and aggrecan expression was increased. When the Notch signaling pathway of ADMSCs was inhibited, the ability of BMP9 overexpressing ADMSCs to repair the cartilage in the OS-affected knee joint was attenuated. These results demonstrate that upregulating BMP9 protein expression may promote the chondrogenic differentiation of ADMSCs. Intra-articular injection of ADMSCs contributes to cartilage repair in OS-affected knee joints through the Notch1/Jagged1 signaling pathway.
Keywords: BMP9; Notch1/Jagged1 signaling pathway; adipose-derived mesenchymal stem cells; osteoarthritis.
Figures

Similar articles
-
Human adipose and synovial mesenchymal stem cells improve osteoarthritis in rats by reducing chondrocyte reactive oxygen species and inhibiting inflammatory response.J Clin Lab Anal. 2022 May;36(5):e24353. doi: 10.1002/jcla.24353. Epub 2022 Mar 21. J Clin Lab Anal. 2022. PMID: 35312120 Free PMC article.
-
Adipose derived mesenchymal stem cells alleviated osteoarthritis and chondrocyte apoptosis through autophagy inducing.J Cell Biochem. 2019 Feb;120(2):2198-2212. doi: 10.1002/jcb.27530. Epub 2018 Oct 13. J Cell Biochem. 2019. PMID: 30315711
-
Effects of Combined Allogenic Adipose Stem Cells and Hyperbaric Oxygenation Treatment on Pathogenesis of Osteoarthritis in Knee Joint Induced by Monoiodoacetate.Int J Mol Sci. 2022 Jul 12;23(14):7695. doi: 10.3390/ijms23147695. Int J Mol Sci. 2022. PMID: 35887046 Free PMC article.
-
The role of adipose-derived mesenchymal stem cells in knee osteoarthritis: a meta-analysis of randomized controlled trials.Ther Adv Musculoskelet Dis. 2022 Dec 26;14:1759720X221146005. doi: 10.1177/1759720X221146005. eCollection 2022. Ther Adv Musculoskelet Dis. 2022. PMID: 36601089 Free PMC article.
-
Chondrogenic differentiation of mesenchymal stem cells and its clinical applications.Yonsei Med J. 2004 Jun 30;45 Suppl:41-7. doi: 10.3349/ymj.2004.45.Suppl.41. Yonsei Med J. 2004. PMID: 15250049 Review.
Cited by
-
Acupuncture for Knee Osteoarthritis: A Systematic Review of Randomized Clinical Trials with Meta-Analyses and Trial Sequential Analyses.Biomed Res Int. 2022 Apr 21;2022:6561633. doi: 10.1155/2022/6561633. eCollection 2022. Biomed Res Int. 2022. Retraction in: Biomed Res Int. 2023 Nov 29;2023:9824061. doi: 10.1155/2023/9824061. PMID: 35496051 Free PMC article. Retracted.
-
The role of the immune microenvironment in bone, cartilage, and soft tissue regeneration: from mechanism to therapeutic opportunity.Mil Med Res. 2022 Nov 19;9(1):65. doi: 10.1186/s40779-022-00426-8. Mil Med Res. 2022. PMID: 36401295 Free PMC article. Review.
-
The importance of BMPs and TGF-βs for endochondral bone repair - A longitudinal study in hip arthroplasty patients.Bone Rep. 2023 Nov 10;19:101723. doi: 10.1016/j.bonr.2023.101723. eCollection 2023 Dec. Bone Rep. 2023. PMID: 38047271 Free PMC article.
-
The potential regulatory role of BMP9 in inflammatory responses.Genes Dis. 2021 Sep 21;9(6):1566-1578. doi: 10.1016/j.gendis.2021.08.010. eCollection 2022 Nov. Genes Dis. 2021. PMID: 36157503 Free PMC article. Review.
-
Indocyanine Green Uptake and Periodic Acid-Schiff Staining Method for Function Detection of Liver Cells are Affected by Different Cell Confluence.Cytotechnology. 2021 Apr;73(2):159-167. doi: 10.1007/s10616-021-00453-8. Epub 2021 Jan 30. Cytotechnology. 2021. PMID: 33927473 Free PMC article.
References
-
- Ding S, Zheng K. Artificial total hip arthroplasty with collum femoris preserving for treating hip joint. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2010;24:1–4. (In Chinese) - PubMed
-
- Komaki M, Iwasaki K, Morita I. Bone and stem cells. Mesenchymal stem cells and bone regeneration. Clin Calcium. 2014;24:565–573. - PubMed
-
- Endo I, Mastumoto T. Bone and stem cells. Regulatory mechanism of mesenchymal stem cell differentiation to osteoblasts. Clin Calcium. 2014;24:555–564. - PubMed
LinkOut - more resources
Full Text Sources
