Age-Dependent Protection of Insulin Secretion in Diet Induced Obese Mice
- PMID: 30546031
- PMCID: PMC6292902
- DOI: 10.1038/s41598-018-36289-0
Age-Dependent Protection of Insulin Secretion in Diet Induced Obese Mice
Abstract
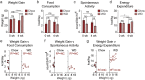
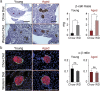
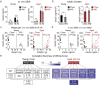
Type 2 diabetes is an age-and-obesity associated disease driven by impairments in glucose homeostasis that ultimately result in defective insulin secretion from pancreatic β-cells. To deconvolve the effects of age and obesity in an experimental model of prediabetes, we fed young and aged mice either chow or a short-term high-fat/high-sucrose Western diet (WD) and examined how weight, glucose tolerance, and β-cell function were affected. Although WD induced a similar degree of weight gain in young and aged mice, a high degree of heterogeneity was found exclusively in aged mice. Weight gain in WD-fed aged mice was well-correlated with glucose intolerance, fasting insulin, and in vivo glucose-stimulated insulin secretion, relationships that were not observed in young animals. Although β-cell mass expansion in the WD-fed aged mice was only three-quarters of that observed in young mice, the islets from aged mice were resistant to the sharp WD-induced decline in ex vivo insulin secretion observed in young mice. Our findings demonstrate that age is associated with the protection of islet function in diet-induced obese mice, and furthermore, that WD challenge exposes variability in the resilience of the insulin secretory pathway in aged mice.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Taurine supplementation prevents morpho-physiological alterations in high-fat diet mice pancreatic β-cells.Amino Acids. 2012 Oct;43(4):1791-801. doi: 10.1007/s00726-012-1263-5. Epub 2012 Mar 15. Amino Acids. 2012. PMID: 22418865
-
Duodenal-jejunal bypass surgery enhances glucose tolerance and beta-cell function in Western diet obese rats.Obes Surg. 2012 May;22(5):819-26. doi: 10.1007/s11695-012-0630-3. Obes Surg. 2012. PMID: 22411572
-
Amelioration of high fat diet-induced glucose intolerance by blockade of Smad4 in pancreatic beta-cells.Exp Clin Endocrinol Diabetes. 2015 Apr;123(4):221-6. doi: 10.1055/s-0034-1395583. Epub 2014 Dec 11. Exp Clin Endocrinol Diabetes. 2015. PMID: 25502579
-
Metallothionein 1 negatively regulates glucose-stimulated insulin secretion and is differentially expressed in conditions of beta cell compensation and failure in mice and humans.Diabetologia. 2019 Dec;62(12):2273-2286. doi: 10.1007/s00125-019-05008-3. Epub 2019 Oct 17. Diabetologia. 2019. PMID: 31624901
-
[Insulin secretion and insulin resistance].Nihon Rinsho. 2013 Nov;71(11):1936-40. Nihon Rinsho. 2013. PMID: 24397162 Review. Japanese.
Cited by
-
G6PC2 confers protection against hypoglycemia upon ketogenic diet feeding and prolonged fasting.Mol Metab. 2020 Nov;41:101043. doi: 10.1016/j.molmet.2020.101043. Epub 2020 Jun 20. Mol Metab. 2020. PMID: 32569842 Free PMC article.
-
Redox homeostasis and cell cycle activation mediate beta-cell mass expansion in aged, diabetes-prone mice under metabolic stress conditions: Role of thioredoxin-interacting protein (TXNIP).Redox Biol. 2020 Oct;37:101748. doi: 10.1016/j.redox.2020.101748. Epub 2020 Oct 7. Redox Biol. 2020. PMID: 33128997 Free PMC article.
-
A toolbox for the longitudinal assessment of healthspan in aging mice.Nat Protoc. 2020 Feb;15(2):540-574. doi: 10.1038/s41596-019-0256-1. Epub 2020 Jan 8. Nat Protoc. 2020. PMID: 31915391 Free PMC article.
-
Synthesis and Biological Validation of a Harmine-Based, Central Nervous System (CNS)-Avoidant, Selective, Human β-Cell Regenerative Dual-Specificity Tyrosine Phosphorylation-Regulated Kinase A (DYRK1A) Inhibitor.J Med Chem. 2020 Mar 26;63(6):2986-3003. doi: 10.1021/acs.jmedchem.9b01379. Epub 2020 Feb 19. J Med Chem. 2020. PMID: 32003560 Free PMC article.
-
Myeloperoxidase, extracellular DNA and neutrophil extracellular trap formation in the animal models of metabolic dysfunction-associated steatotic liver disease.World J Gastroenterol. 2025 Jul 21;31(27):106166. doi: 10.3748/wjg.v31.i27.106166. World J Gastroenterol. 2025. PMID: 40741100 Free PMC article.
References
-
- National Diabetes Report Card. (Center for Disease Control, 2017).
-
- National Diabetes Statistics Report. (Center for Disease Control, 2018).
Publication types
MeSH terms
Substances
Grants and funding
- AG051974/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- DK113103/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- R21 AG050135/AG/NIA NIH HHS/United States
- R01 AG056771/AG/NIA NIH HHS/United States
- DK101683/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- R21 AG051974/AG/NIA NIH HHS/United States
- K01 DK101683/DK/NIDDK NIH HHS/United States
- R01 DK102598/DK/NIDDK NIH HHS/United States
- AG041765/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- F31 DK109698/DK/NIDDK NIH HHS/United States
- 1-16-IBS-212/American Diabetes Association (ADA)/International
- AG050135/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- I01 BX004031/BX/BLRD VA/United States
- T32GM008349/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- R01 DK113103/DK/NIDDK NIH HHS/United States
- DK102598/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
- R00 AG041765/AG/NIA NIH HHS/United States
- T32 AG000213/AG/NIA NIH HHS/United States
- AG056771/U.S. Department of Health & Human Services | National Institutes of Health (NIH)/International
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

