Timed Collinear Activation of Hox Genes during Gastrulation Controls the Avian Forelimb Position
- PMID: 30554902
- PMCID: PMC6331352
- DOI: 10.1016/j.cub.2018.11.009
Timed Collinear Activation of Hox Genes during Gastrulation Controls the Avian Forelimb Position
Abstract
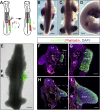
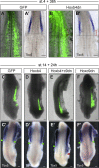
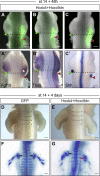
Limb position along the body is highly consistent within one species but very variable among vertebrates. Despite major advances in our understanding of limb patterning in three dimensions, how limbs reproducibly form along the antero-posterior axis remains largely unknown. Hox genes have long been suspected to control limb position; however, supporting evidences are mostly correlative and their role in this process is unclear. Here, we show that limb position is determined early in development through the action of Hox genes. Dynamic lineage analysis revealed that, during gastrulation, the forelimb, interlimb, and hindlimb fields are progressively generated and concomitantly patterned by the collinear activation of Hox genes in a two-step process. First, the sequential activation of Hoxb genes controls the relative position of their own collinear domains of expression in the forming lateral plate mesoderm, as demonstrated by functional perturbations during gastrulation. Then, within these collinear domains, we show that Hoxb4 anteriorly and Hox9 genes posteriorly, respectively, activate and repress the expression of the forelimb initiation gene Tbx5 and instruct the definitive position of the forelimb. Furthermore, by comparing the dynamics of Hoxb genes activation during zebra finch, chicken, and ostrich gastrulation, we provide evidences that changes in the timing of collinear Hox gene activation might underlie natural variation in forelimb position between different birds. Altogether, our results that characterize the cellular and molecular mechanisms underlying the regulation and natural variation of forelimb positioning in avians show a direct and early role for Hox genes in this process.
Keywords: Hox genes; Tbx5; birds; chicken; collinearity; gastrulation; lateral plate mesoderm; limb; patterning.
Copyright © 2018 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Figures




Comment in
-
Developmental Biology: Hox Timing Determines Limb Placement.Curr Biol. 2019 Jan 21;29(2):R52-R54. doi: 10.1016/j.cub.2018.11.068. Curr Biol. 2019. PMID: 30668947
Similar articles
-
A combination of activation and repression by a colinear Hox code controls forelimb-restricted expression of Tbx5 and reveals Hox protein specificity.PLoS Genet. 2014 Mar 20;10(3):e1004245. doi: 10.1371/journal.pgen.1004245. eCollection 2014 Mar. PLoS Genet. 2014. PMID: 24651482 Free PMC article.
-
Hox genes regulate the onset of Tbx5 expression in the forelimb.Development. 2012 Sep;139(17):3180-8. doi: 10.1242/dev.084814. Development. 2012. PMID: 22872086 Free PMC article.
-
Developmental Biology: Hox Timing Determines Limb Placement.Curr Biol. 2019 Jan 21;29(2):R52-R54. doi: 10.1016/j.cub.2018.11.068. Curr Biol. 2019. PMID: 30668947
-
Subdivision of the lateral plate mesoderm and specification of the forelimb and hindlimb forming domains.Semin Cell Dev Biol. 2016 Jan;49:102-8. doi: 10.1016/j.semcdb.2015.11.011. Epub 2015 Nov 28. Semin Cell Dev Biol. 2016. PMID: 26643124 Review.
-
How the embryo makes a limb: determination, polarity and identity.J Anat. 2015 Oct;227(4):418-30. doi: 10.1111/joa.12361. Epub 2015 Aug 7. J Anat. 2015. PMID: 26249743 Free PMC article. Review.
Cited by
-
The lateral plate mesoderm.Development. 2020 Jun 19;147(12):dev175059. doi: 10.1242/dev.175059. Development. 2020. PMID: 32561665 Free PMC article. Review.
-
Retinoic acid influences the timing and scaling of avian wing development.Cell Rep. 2022 Jan 25;38(4):110288. doi: 10.1016/j.celrep.2021.110288. Cell Rep. 2022. PMID: 35081337 Free PMC article.
-
Transgenesis and web resources in quail.Elife. 2020 May 27;9:e56312. doi: 10.7554/eLife.56312. Elife. 2020. PMID: 32459172 Free PMC article.
-
Novel function of Hox13 in regulating outgrowth of the newt hindlimb bud through interaction with Fgf10 and Tbx4.Dev Growth Differ. 2025 Jan;67(1):10-22. doi: 10.1111/dgd.12952. Epub 2024 Dec 26. Dev Growth Differ. 2025. PMID: 39725403 Free PMC article.
-
Homeotic and nonhomeotic patterns in the tetrapod vertebral formula.Proc Natl Acad Sci U S A. 2024 Nov 19;121(47):e2411421121. doi: 10.1073/pnas.2411421121. Epub 2024 Nov 11. Proc Natl Acad Sci U S A. 2024. PMID: 39527744 Free PMC article.
References
-
- Tanaka M. Molecular and evolutionary basis of limb field specification and limb initiation. Dev. Growth Differ. 2013;55:149–163. - PubMed
-
- Dollé P., Izpisúa-Belmonte J.-C., Falkenstein H., Renucci A., Duboule D. Coordinate expression of the murine Hox-5 complex homoeobox-containing genes during limb pattern formation. Nature. 1989;342:767–772. - PubMed
-
- Gaunt S.J., Sharpe P.T., Duboule D. Spatially restricted domains of homeo-gene transcripts in mouse embryos: relation to a segmented body plan. Development. 1988;104:169–179.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials

