A monoclonal antibody-based urine Histoplasma antigen enzyme immunoassay (IMMY®) for the diagnosis of histoplasmosis in cats
- PMID: 30557457
- PMCID: PMC6430878
- DOI: 10.1111/jvim.15379
A monoclonal antibody-based urine Histoplasma antigen enzyme immunoassay (IMMY®) for the diagnosis of histoplasmosis in cats
Abstract
Background: An in-house Histoplasma urine antigen test for cats might be desirable in certain situations.
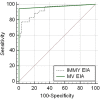
Objective: To validate and compare the diagnostic performance of a monoclonal antibody-based IMMY urine Histoplasma antigen enzyme immunoassay (IMMY EIA) to the commercially available urine Histoplasma antigen enzyme immunoassay (MiraVista Diagnostics, MV EIA).
Animals: One hundred ninety-three urine samples from 105 client-owned and purpose-bred research cats.
Methods: Cats were classified as Histoplasma positive or negative based on diagnostic investigation. The IMMY EIA and MV EIA were performed on all urine samples. Correlation and agreement between the assays were determined. Diagnostic performance was determined and compared between assays.
Results: The IMMY EIA, with a 0.25 ng/mL diagnostic cutoff, provided a diagnostic sensitivity (DSe), diagnostic specificity (DSp), and diagnostic accuracy (DAc) of 89% (95% confidence interval [CI]; 73%-97%), 80% (67%-89%), and 83% (74%-90%), respectively. The IMMY EIA, with a 1.1 ng/mL diagnostic cutoff, provided a DSe, DSp, and DAc of 77% (95% CI 60%-90%), 97% (88%-100%), and 89% (81%-95%), respectively. The MV EIA provided a DSe, DSp, and DAc of 94% (95% CI 81%-99%), 97% (89%-100%), and 96% (90%-99%), respectively. Moderate overall agreement was found between MV EIA and IMMY EIA using the 0.25 ng/mL cut-off (к = 0.44; 95% CI 0.31-0.57) and the 1.1 ng/mL cut-off (к = 0.43, 95% CI, 0.31-0.56).
Conclusions and clinical importance: The IMMY EIA might be useful as a diagnostic test for histoplasmosis in cats. Further modifications of the IMMY EIA are required to achieve the diagnostic performance of the MV EIA.
Keywords: feline; fungal; histoplasmosis; mycosis.
© 2018 The Authors. Journal of Veterinary Internal Medicine published by Wiley Periodicals, Inc. on behalf of the American College of Veterinary Internal Medicine.
Conflict of interest statement
Dr. Andrew Hanzlicek has in the past, and is currently, collaborating with MiraVista Diagnostics on multiple studies relating to veterinary clinical mycology.
Figures


Similar articles
-
Pretreatment Histoplasma capsulatum urine enzymatic immunoassay concentrations do not correlate with outcome but may be influenced by renal function in cats with histoplasmosis.J Feline Med Surg. 2018 Dec;20(12):1177-1179. doi: 10.1177/1098612X18761440. Epub 2018 Mar 7. J Feline Med Surg. 2018. PMID: 29513156 Free PMC article.
-
Evaluation of the IMMY ALPHA Histoplasma antigen enzyme immunoassay for diagnosis of histoplasmosis marked by antigenuria.Clin Vaccine Immunol. 2007 Jun;14(6):802-3. doi: 10.1128/CVI.00035-07. Epub 2007 Apr 4. Clin Vaccine Immunol. 2007. PMID: 17409220 Free PMC article.
-
Evaluation of a novel monoclonal antibody-based enzyme immunoassay for detection of Histoplasma antigen in urine of dogs.J Vet Intern Med. 2021 Jan;35(1):284-293. doi: 10.1111/jvim.16006. Epub 2020 Dec 24. J Vet Intern Med. 2021. PMID: 33368653 Free PMC article.
-
Antigen Detection in the Diagnosis of Histoplasmosis: A Meta-analysis of Diagnostic Performance.Mycopathologia. 2016 Apr;181(3-4):197-205. doi: 10.1007/s11046-015-9965-3. Epub 2015 Nov 11. Mycopathologia. 2016. PMID: 26559429 Review.
-
Histoplasma capsulatum antigen detection tests as an essential diagnostic tool for patients with advanced HIV disease in low and middle income countries: A systematic review of diagnostic accuracy studies.PLoS Negl Trop Dis. 2018 Oct 19;12(10):e0006802. doi: 10.1371/journal.pntd.0006802. eCollection 2018 Oct. PLoS Negl Trop Dis. 2018. PMID: 30339674 Free PMC article.
Cited by
-
Timely Diagnosis of Histoplasmosis in Non-endemic Countries: A Laboratory Challenge.Front Microbiol. 2020 Mar 24;11:467. doi: 10.3389/fmicb.2020.00467. eCollection 2020. Front Microbiol. 2020. PMID: 32269555 Free PMC article. Review.
-
The Association of Chronic Pulmonary Aspergillosis and Chronic Pulmonary Histoplasmosis with MDR-TB Patients in Indonesia.J Fungi (Basel). 2024 Jul 29;10(8):529. doi: 10.3390/jof10080529. J Fungi (Basel). 2024. PMID: 39194855 Free PMC article.
-
Diagnosis of histoplasmosis: current status and perspectives.Appl Microbiol Biotechnol. 2021 Mar;105(5):1837-1859. doi: 10.1007/s00253-021-11170-9. Epub 2021 Feb 15. Appl Microbiol Biotechnol. 2021. PMID: 33587157 Review.
-
Clinical utility of fungal culture and antifungal susceptibility in cats and dogs with histoplasmosis.J Vet Intern Med. 2023 May-Jun;37(3):998-1006. doi: 10.1111/jvim.16725. Epub 2023 Apr 24. J Vet Intern Med. 2023. PMID: 37092675 Free PMC article.
-
Evaluation of an enzyme immunoassay and immunodiffusion for detection of anti-Histoplasma antibodies in serum from cats and dogs.J Vet Intern Med. 2023 May-Jun;37(3):1007-1014. doi: 10.1111/jvim.16726. Epub 2023 Apr 27. J Vet Intern Med. 2023. PMID: 37102374 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

