Study of New Therapeutic Strategies to Combat Breast Cancer Using Drug Combinations
- PMID: 30558247
- PMCID: PMC6315516
- DOI: 10.3390/biom8040175
Study of New Therapeutic Strategies to Combat Breast Cancer Using Drug Combinations
Abstract
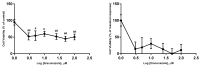
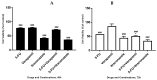
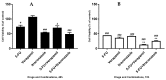
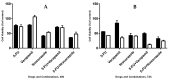
Cancer is a disease that affects and kills millions of people worldwide. Breast cancer, especially, has a high incidence and mortality, and is challenging to treat. Due to its high impact on the health sector, oncological therapy is the subject of an intense and very expensive research. To improve this therapy and reduce its costs, strategies such as drug repurposing and drug combinations have been extensively studied. Drug repurposing means giving new usefulness to drugs which are approved for the therapy of various diseases, but, in this case, are not approved for cancer therapy. On the other hand, the purpose of combining drugs is that the response that is obtained is more advantageous than the response obtained by the single drugs. Using drugs with potential to be repurposed, combined with 5-fluorouracil, the aim of this project was to investigate whether this combination led to therapeutic benefits, comparing with the isolated drugs. We started with a screening of the most promising drugs, with verapamil and itraconazole being chosen. Several cellular viability studies, cell death and proliferation studies, mainly in MCF-7 cells (Michigan Cancer Foundation-7, human breast adenocarcinoma cells) were performed. Studies were also carried out to understand the effect of the drugs at the level of possible therapeutic resistance, evaluating the epithelial-mesenchymal transition. Combining all the results, the conclusion is that the combination of verapamil and itraconazole with 5-fluorouracil had benefits, mainly by decreasing cell viability and proliferation. Furthermore, the combination of itraconazole and 5-fluorouracil seemed to be the most effective, being an interesting focus in future studies.
Keywords: 5-fluorouracil; breast cancer; cell viability; drug combinations; drug repurposing; epithelial-mesenchymal transition.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



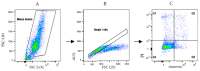
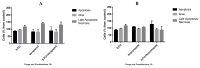
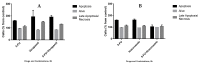
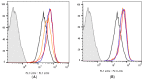

Similar articles
-
Drug Combinations: A New Strategy to Extend Drug Repurposing and Epithelial-Mesenchymal Transition in Breast and Colon Cancer Cells.Biomolecules. 2022 Jan 23;12(2):190. doi: 10.3390/biom12020190. Biomolecules. 2022. PMID: 35204691 Free PMC article.
-
The YB-1/EZH2/amphiregulin signaling axis mediates LPA-induced breast cancer cell invasion.Arch Pharm Res. 2019 Jun;42(6):519-530. doi: 10.1007/s12272-019-01149-6. Epub 2019 Apr 19. Arch Pharm Res. 2019. PMID: 31004257
-
Epithelial to mesenchymal transition is associated with rapamycin resistance.Oncotarget. 2015 Aug 14;6(23):19500-13. doi: 10.18632/oncotarget.3669. Oncotarget. 2015. PMID: 25944619 Free PMC article.
-
Drug combination and repurposing for cancer therapy: the example of breast cancer.Heliyon. 2021 Jan 11;7(1):e05948. doi: 10.1016/j.heliyon.2021.e05948. eCollection 2021 Jan. Heliyon. 2021. PMID: 33490692 Free PMC article. Review.
-
Platinum compounds in the treatment of advanced breast cancer.Clin Breast Cancer. 2001 Oct;2(3):190-208; discussion 209. doi: 10.3816/CBC.2001.n.022. Clin Breast Cancer. 2001. PMID: 11899413 Review.
Cited by
-
Role of Hedgehog Signaling in Breast Cancer: Pathogenesis and Therapeutics.Cells. 2019 Apr 25;8(4):375. doi: 10.3390/cells8040375. Cells. 2019. PMID: 31027259 Free PMC article. Review.
-
High Drug Resistance in Feline Mammary Carcinoma Cell Line (FMCm) and Comparison with Human Breast Cancer Cell Line (MCF-7).Animals (Basel). 2021 Aug 6;11(8):2321. doi: 10.3390/ani11082321. Animals (Basel). 2021. PMID: 34438778 Free PMC article.
-
Multi-Targeted Anticancer Activity of Imidazolate Phosphane Gold(I) Compounds by Inhibition of DHFR and TrxR in Breast Cancer Cells.Front Chem. 2021 Jan 11;8:602845. doi: 10.3389/fchem.2020.602845. eCollection 2020. Front Chem. 2021. PMID: 33490036 Free PMC article.
-
Synthetic Peptides Induce Human Colorectal Cancer Cell Death via Proapoptotic Pathways.ACS Omega. 2024 Oct 11;9(42):43252-43263. doi: 10.1021/acsomega.4c08194. eCollection 2024 Oct 22. ACS Omega. 2024. PMID: 39464451 Free PMC article.
-
Enhancing the in vitro and in vivo activity of itraconazole against breast cancer using miltefosine-modified lipid nanocapsules.Drug Deliv. 2021 Dec;28(1):906-919. doi: 10.1080/10717544.2021.1917728. Drug Deliv. 2021. PMID: 33960245 Free PMC article.
References
-
- Malani P.N. Harrison’s Principles of Internal Medicine. JAMA. 2012;308:1813–1814. doi: 10.1001/jama.308.17.1813-b. - DOI
-
- Cancer Research UK Worldwide Cancer Statistics. [(accessed on 8 August 2018)]; Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/w....
-
- Cancer Therapy Advisor Breast Cancer (Invasive) Treatment Regimens. [(accessed on 21 March 2018)]; Available online: https://www.cancertherapyadvisor.com/breast-cancer/breast-cancer-invasiv...
-
- Green R.J., Harris N.D. Pathology and Therapeutics for Pharmacists. 3rd ed. Pharmaceutical Press; London, UK: 2008.
-
- Dawood S., Austin L., Cristofanilli M. Cancer stem cells: Implications for cancer therapy. Oncology (Williston Park) 2014;28:1101–1107, 1110. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

