Quantitative Assessment of rPM6 for Fluorine- and Chlorine-Containing Metal Complexes: Comparison with Experimental, First-Principles, and Other Semiempirical Results
- PMID: 30558286
- PMCID: PMC6321459
- DOI: 10.3390/molecules23123332
Quantitative Assessment of rPM6 for Fluorine- and Chlorine-Containing Metal Complexes: Comparison with Experimental, First-Principles, and Other Semiempirical Results
Abstract
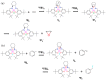
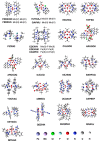
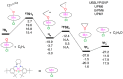
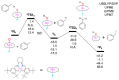
We report a reparameterization of PM6 parameters for fluorine and chlorine using our training set containing transition metal complexes. Spin unrestricted calculations with the resulting rPM6 (UrPM6) were examined quantitatively using two test sets: (i) the description of magnetic interactions in 25 dinuclear metal complexes and (ii) the prediction of barrier heights and reaction energies for epoxidation and fluorination reactions catalyzed by high-valent manganese-oxo species. The conventional UPM6 and UPM7 methods were also evaluated for comparison on the basis of either experimental or computational (the UB3LYP/SVP level) outcomes. The merits of UrPM6 are highlighted by both the test sets. As regards magnetic exchange coupling constants, the UrPM6 method had the smallest mean absolute errors from the experimental data (19 cm-1), followed by UPM7 (119 cm-1) and UPM6 (373 cm-1). For the epoxidation and fluorination reactions, all of the transition state searches were successful using UrPM6, while the success rates obtained by UPM6 and UPM7 were only 50%. The UrPM6-optimized stationary points also agreed well with the reference UB3LYP-optimized geometries. The accuracy for estimating reaction energies was also greatly remedied.
Keywords: magnetic exchange coupling; oxidation reaction; semiempirical method.
Conflict of interest statement
The authors declare no conflict of interests.
Figures


References
-
- Zhang W., Loebach J.L., Wilson S.R., Jacobsen E.N. Enantioselective epoxidation of unfunctionalized olefins catalyzed by salen managanese complexes. J. Am. Chem. Soc. 1990;112:2801–2803. doi: 10.1021/ja00163a052. - DOI
-
- Irie R., Noda K., Ito Y., Katsuki T. Enantioselective epoxidation of unfunctionalized olefins using chiral (salen)manganese(III) complexes. Tetrahedron Lett. 1991;32:1055–1058. doi: 10.1016/S0040-4039(00)74486-8. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

