Lenalidomide maintenance versus observation for patients with newly diagnosed multiple myeloma (Myeloma XI): a multicentre, open-label, randomised, phase 3 trial
- PMID: 30559051
- PMCID: PMC6318225
- DOI: 10.1016/S1470-2045(18)30687-9
Lenalidomide maintenance versus observation for patients with newly diagnosed multiple myeloma (Myeloma XI): a multicentre, open-label, randomised, phase 3 trial
Abstract
Background: Patients with multiple myeloma treated with lenalidomide maintenance therapy have improved progression-free survival, primarily following autologous stem-cell transplantation. A beneficial effect of lenalidomide maintenance therapy on overall survival in this setting has been inconsistent between individual studies. Minimal data are available on the effect of maintenance lenalidomide in more aggressive disease states, such as patients with cytogenetic high-risk disease or patients ineligible for transplantation. We aimed to assess lenalidomide maintenance versus observation in patients with newly diagnosed multiple myeloma, including cytogenetic risk and transplantation status subgroup analyses.
Methods: The Myeloma XI trial was an open-label, randomised, phase 3, adaptive design trial with three randomisation stages done at 110 National Health Service hospitals in England, Wales, and Scotland. There were three potential randomisations in the study: induction treatment (allocation by transplantation eligibility status); intensification treatment (allocation by response to induction therapy); and maintenance treatment. Here, we report the results of the randomisation to maintenance treatment. Eligible patients for maintenance randomisation were aged 18 years or older and had symptomatic or non-secretory multiple myeloma, had completed their assigned induction therapy as per protocol and had achieved at least a minimal response to protocol treatment, including lenalidomide. Patients were randomly assigned (1:1 from Jan 13, 2011, to Jun 27, 2013, and 2:1 from Jun 28, 2013, to Aug 11, 2017) to lenalidomide maintenance (10 mg orally on days 1-21 of a 28-day cycle) or observation, and stratified by allocated induction and intensification treatment, and centre. The co-primary endpoints were progression-free survival and overall survival, analysed by intention to treat. Safety analysis was per protocol. This study is registered with the ISRCTN registry, number ISRCTN49407852, and clinicaltrialsregister.eu, number 2009-010956-93, and has completed recruitment.
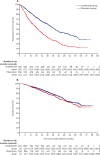
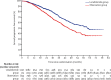
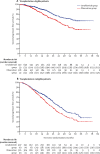
Findings: Between Jan 13, 2011, and Aug 11, 2017, 1917 patients were accrued to the maintenance treatment randomisation of the trial. 1137 patients were assigned to lenalidomide maintenance and 834 patients to observation. After a median follow-up of 31 months (IQR 18-50), median progression-free survival was 39 months (95% CI 36-42) with lenalidomide and 20 months (18-22) with observation (hazard ratio [HR] 0·46 [95% CI 0·41-0·53]; p<0·0001), and 3-year overall survival was 78·6% (95% Cl 75·6-81·6) in the lenalidomide group and 75·8% (72·4-79·2) in the observation group (HR 0·87 [95% CI 0·73-1·05]; p=0·15). Progression-free survival was improved with lenalidomide compared with observation across all prespecified subgroups. On prespecified subgroup analyses by transplantation status, 3-year overall survival in transplantation-eligible patients was 87·5% (95% Cl 84·3-90·7) in the lenalidomide group and 80·2% (76·0-84·4) in the observation group (HR 0·69 [95% CI 0·52-0·93]; p=0·014), and in transplantation-ineligible patients it was 66·8% (61·6-72·1) in the lenalidomide group and 69·8% (64·4-75·2) in the observation group (1·02 [0·80-1·29]; p=0·88). By cytogenetic risk group, in standard-risk patients, 3-year overall survival was 86·4% (95% CI 80·0-90·9) in the lenalidomide group compared with 81·3% (74·2-86·7) in the observation group, and in high-risk patients, it was 74.9% (65·8-81·9) in the lenalidomide group compared with 63·7% (52·8-72·7) in the observation group; and in ultra-high-risk patients it was 62·9% (46·0-75·8) compared with 43·5% (22·2-63·1). Since these subgroup analyses results were not powered they should be interpreted with caution. The most common grade 3 or 4 adverse events for patients taking lenalidomide were haematological, including neutropenia (362 [33%] patients), thrombocytopenia (72 [7%] patients), and anaemia (42 [4%] patients). Serious adverse events were reported in 494 (45%) of 1097 patients receiving lenalidomide compared with 150 (17%) of 874 patients on observation. The most common serious adverse events were infections in both the lenalidomide group and the observation group. 460 deaths occurred during maintenance treatment, 234 (21%) in the lenalidomide group and 226 (27%) in the observation group, and no deaths in the lenalidomide group were deemed treatment related.
Interpretation: Maintenance therapy with lenalidomide significantly improved progression-free survival in patients with newly diagnosed multiple myeloma compared with observation, but did not improve overall survival in the intention-to-treat analysis of the whole trial population. The manageable safety profile of this drug and the encouraging results in subgroup analyses of patients across all cytogenetic risk groups support further investigation of maintenance lenalidomide in this setting.
Funding: Cancer Research UK, Celgene, Amgen, Merck, and Myeloma UK.
Copyright © 2019 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Figures




Comment in
-
Lenalidomide as maintenance for every newly diagnosed patient with multiple myeloma.Lancet Oncol. 2019 Jan;20(1):5-6. doi: 10.1016/S1470-2045(18)30764-2. Epub 2018 Dec 14. Lancet Oncol. 2019. PMID: 30559052 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

