Persistent arterial wall inflammation in patients with elevated lipoprotein(a) despite strong low-density lipoprotein cholesterol reduction by proprotein convertase subtilisin/kexin type 9 antibody treatment
- PMID: 30561610
- PMCID: PMC6933872
- DOI: 10.1093/eurheartj/ehy862
Persistent arterial wall inflammation in patients with elevated lipoprotein(a) despite strong low-density lipoprotein cholesterol reduction by proprotein convertase subtilisin/kexin type 9 antibody treatment
Abstract
Aims: Subjects with lipoprotein(a) [Lp(a)] elevation have increased arterial wall inflammation and cardiovascular risk. In patients at increased cardiovascular risk, arterial wall inflammation is reduced following lipid-lowering therapy by statin treatment or lipoprotein apheresis. However, it is unknown whether lipid-lowering treatment in elevated Lp(a) subjects alters arterial wall inflammation. We evaluated whether evolocumab, which lowers both low-density lipoprotein cholesterol (LDL-C) and Lp(a), attenuates arterial wall inflammation in patients with elevated Lp(a).
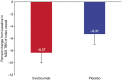
Methods and results: In this multicentre, randomized, double-blind, placebo-controlled study, 129 patients {median [interquartile range (IQR)]: age 60.0 [54.0-67.0] years, Lp(a) 200.0 [155.5-301.5] nmol/L [80.0 (62.5-121.0) mg/dL]; mean [standard deviation (SD)] LDL-C 3.7 [1.0] mmol/L [144.0 (39.7) mg/dL]; National Cholesterol Education Program high risk, 25.6%} were randomized to monthly subcutaneous evolocumab 420 mg or placebo. Compared with placebo, evolocumab reduced LDL-C by 60.7% [95% confidence interval (CI) 65.8-55.5] and Lp(a) by 13.9% (95% CI 19.3-8.5). Among evolocumab-treated patients, the Week 16 mean (SD) LDL-C level was 1.6 (0.7) mmol/L [60.1 (28.1) mg/dL], and the median (IQR) Lp(a) level was 188.0 (140.0-268.0) nmol/L [75.2 (56.0-107.2) mg/dL]. Arterial wall inflammation [most diseased segment target-to-background ratio (MDS TBR)] in the index vessel (left carotid, right carotid, or thoracic aorta) was assessed by 18F-fluoro-deoxyglucose positron-emission tomography/computed tomography. Week 16 index vessel MDS TBR was not significantly altered with evolocumab (-8.3%) vs. placebo (-5.3%) [treatment difference -3.0% (95% CI -7.4% to 1.4%); P = 0.18].
Conclusion: Evolocumab treatment in patients with median baseline Lp(a) 200.0 nmol/L led to a large reduction in LDL-C and a small reduction in Lp(a), resulting in persistent elevated Lp(a) levels. The latter may have contributed to the unaltered arterial wall inflammation.
Keywords: Arterial wall inflammation; Atherosclerosis; Evolocumab; Lipoprotein(a); PCSK9 antibodies.
Published on behalf of the European Society of Cardiology. All rights reserved. © The Author(s) 2018. For permissions, please email: journals.permissions@oup.com.
Figures



Comment in
-
Lipoprotein (a), arterial inflammation, and PCSK9 inhibition.Eur Heart J. 2019 Sep 1;40(33):2782-2784. doi: 10.1093/eurheartj/ehz087. Eur Heart J. 2019. PMID: 30838412 No abstract available.
Similar articles
-
Lipoprotein(a), PCSK9 Inhibition, and Cardiovascular Risk.Circulation. 2019 Mar 19;139(12):1483-1492. doi: 10.1161/CIRCULATIONAHA.118.037184. Circulation. 2019. PMID: 30586750 Clinical Trial.
-
Relationship Between Low-Density Lipoprotein Cholesterol and Lipoprotein(a) Lowering in Response to PCSK9 Inhibition With Evolocumab.J Am Heart Assoc. 2019 Feb 19;8(4):e010932. doi: 10.1161/JAHA.118.010932. J Am Heart Assoc. 2019. PMID: 30755061 Free PMC article. Clinical Trial.
-
Metabolic effects of PCSK9 inhibition with Evolocumab in subjects with elevated Lp(a).Lipids Health Dis. 2020 May 11;19(1):91. doi: 10.1186/s12944-020-01280-0. Lipids Health Dis. 2020. PMID: 32393252 Free PMC article. Clinical Trial.
-
Current insights into the German lipoprotein apheresis standard: PCSK9-inhibitors, lipoprotein apheresis or both?Atheroscler Suppl. 2017 Nov;30:44-49. doi: 10.1016/j.atherosclerosissup.2017.05.005. Epub 2017 Jun 1. Atheroscler Suppl. 2017. PMID: 29096860 Review.
-
Lipoprotein(a) Reduction With Proprotein Convertase Subtilisin/Kexin Type 9 Inhibitors: A Systematic Review and Meta-analysis.J Cardiovasc Pharmacol. 2021 Mar 1;77(3):397-407. doi: 10.1097/FJC.0000000000000963. J Cardiovasc Pharmacol. 2021. PMID: 33298738
Cited by
-
PCSK9 as an Atherothrombotic Risk Factor.Int J Mol Sci. 2023 Jan 19;24(3):1966. doi: 10.3390/ijms24031966. Int J Mol Sci. 2023. PMID: 36768292 Free PMC article. Review.
-
The role of inflammation and the possibilities of inflammation reduction to prevent cardiovascular events.Eur Heart J Open. 2022 Jun 14;2(4):oeac039. doi: 10.1093/ehjopen/oeac039. eCollection 2022 Jul. Eur Heart J Open. 2022. PMID: 35919577 Free PMC article. Review.
-
Biotechnology Approaches for the Treatment of Dyslipidemia.Cardiovasc Drugs Ther. 2021 Feb;35(1):167-183. doi: 10.1007/s10557-020-07017-6. Cardiovasc Drugs Ther. 2021. PMID: 32519066
-
Scan-rescan measurement repeatability of 18F-FDG PET/MR imaging of vascular inflammation.J Nucl Cardiol. 2022 Aug;29(4):1660-1670. doi: 10.1007/s12350-021-02627-5. Epub 2021 May 27. J Nucl Cardiol. 2022. PMID: 34046803
-
The Future of Lipid-lowering Therapy.J Clin Med. 2019 Jul 23;8(7):1085. doi: 10.3390/jcm8071085. J Clin Med. 2019. PMID: 31340607 Free PMC article. Review.
References
-
- Gencer B, Kronenberg F, Stroes ES, Mach F.. Lipoprotein(a): the revenant. Eur Heart J 2017;38:1553–1560. - PubMed
-
- Tsimikas S. A test in context: lipoprotein(a): diagnosis, prognosis, controversies, and emerging therapies. J Am Coll Cardiol 2017;69:692–711. - PubMed
-
- van der Valk FM, Bekkering S, Kroon J, Yeang C, Van den Bossche J, van Buul JD, Ravandi A, Nederveen AJ, Verberne HJ, Scipione C, Nieuwdorp M, Joosten LA, Netea MG, Koschinsky ML, Witztum JL, Tsimikas S, Riksen NP, Stroes ES.. Oxidized phospholipids on lipoprotein(a) elicit arterial wall inflammation and an inflammatory monocyte response in humans. Circulation 2016;134:611–624. - PMC - PubMed
-
- Viney NJ, van Capelleveen JC, Geary RS, Xia S, Tami JA, Yu RZ, Marcovina SM, Hughes SG, Graham MJ, Crooke RM, Crooke ST, Witztum JL, Stroes ES, Tsimikas S.. Antisense oligonucleotides targeting apolipoprotein(a) in people with raised lipoprotein(a): two randomised, double-blind, placebo-controlled, dose-ranging trials. Lancet 2016;388:2239–2253. - PubMed
-
- The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology and European Atherosclerosis Society. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur Heart J 2016;37:2999–3058. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

