Design and Properties of Ligand-Conjugated Guanine Oligonucleotides for Recovery of Mutated G-Quadruplexes
- PMID: 30563296
- PMCID: PMC6321378
- DOI: 10.3390/molecules23123228
Design and Properties of Ligand-Conjugated Guanine Oligonucleotides for Recovery of Mutated G-Quadruplexes
Abstract
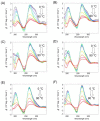
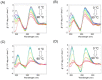
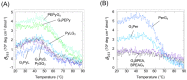
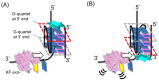
The formation of a guanine quadruplex DNA structure (G4) is known to repress the expression of certain cancer-related genes. Consequently, a mutated G4 sequence can affect quadruplex formation and induce cancer progression. In this study, we developed an oligonucleotide derivative consisting of a ligand-containing guanine tract that replaces the mutated G4 guanine tract at the promoter of the vascular endothelial growth factor (VEGF) gene. A ligand moiety consisting of three types of polyaromatic hydrocarbons, pyrene, anthracene, and perylene, was attached to either the 3' or 5' end of the guanine tract. Each of the ligand-conjugated guanine tracts, with the exception of anthracene derivatives, combined with other intact guanine tracts to form an intermolecular G4 on the mutated VEGF promoter. This intermolecular G4, exhibiting parallel topology and high thermal stability, enabled VEGF G4 formation to be recovered from the mutated sequence. Stability of the intramolecular G4 increased with the size of the conjugated ligand. However, suppression of intermolecular G4 replication was uniquely dependent on whether the ligand was attached to the 3' or 5' end of the guanine tract. These results indicate that binding to either the top or bottom guanine quartet affects unfolding kinetics due to polarization in DNA polymerase processivity. Our findings provide a novel strategy for recovering G4 formation in case of damage, and fine-tuning processes such as replication and transcription.
Keywords: G-quadruplex; cancer; ligand; replication.
Figures




Similar articles
-
Recovery of the Formation and Function of Oxidized G-Quadruplexes by a Pyrene-Modified Guanine Tract.J Am Chem Soc. 2018 May 2;140(17):5774-5783. doi: 10.1021/jacs.8b01577. Epub 2018 Apr 9. J Am Chem Soc. 2018. PMID: 29608858
-
Facilitation of a structural transition in the polypurine/polypyrimidine tract within the proximal promoter region of the human VEGF gene by the presence of potassium and G-quadruplex-interactive agents.Nucleic Acids Res. 2005 Oct 20;33(18):6070-80. doi: 10.1093/nar/gki917. Print 2005. Nucleic Acids Res. 2005. PMID: 16239639 Free PMC article.
-
Chemical Modulation of DNA Replication along G-Quadruplex Based on Topology-Dependent Ligand Binding.J Am Chem Soc. 2021 Oct 13;143(40):16458-16469. doi: 10.1021/jacs.1c05468. Epub 2021 Sep 23. J Am Chem Soc. 2021. PMID: 34554731
-
G-quadruplex DNA: a novel target for drug design.Cell Mol Life Sci. 2021 Oct;78(19-20):6557-6583. doi: 10.1007/s00018-021-03921-8. Epub 2021 Aug 30. Cell Mol Life Sci. 2021. PMID: 34459951 Free PMC article. Review.
-
G-Quadruplex Structures in Bacteria: Biological Relevance and Potential as an Antimicrobial Target.J Bacteriol. 2021 Jun 8;203(13):e0057720. doi: 10.1128/JB.00577-20. Epub 2021 Jun 8. J Bacteriol. 2021. PMID: 33649149 Free PMC article. Review.
Cited by
-
Enzymatic bypass of G-quadruplex structures containing oxidative lesions.Nucleic Acids Res. 2025 Jan 7;53(1):gkae1157. doi: 10.1093/nar/gkae1157. Nucleic Acids Res. 2025. PMID: 39673512 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

