Cardiac rehabilitation to improve health-related quality of life following trans-catheter aortic valve implantation: a randomised controlled feasibility study: RECOVER-TAVI Pilot, ORCA 4, For the Optimal Restoration of Cardiac Activity Group
- PMID: 30564436
- PMCID: PMC6293531
- DOI: 10.1186/s40814-018-0363-8
Cardiac rehabilitation to improve health-related quality of life following trans-catheter aortic valve implantation: a randomised controlled feasibility study: RECOVER-TAVI Pilot, ORCA 4, For the Optimal Restoration of Cardiac Activity Group
Abstract
Objectives: Transcatheter aortic valve implantation (TAVI) is often undertaken in the oldest frailest cohort of patients undergoing cardiac interventions. We plan to investigate the potential benefit of cardiac rehabilitation (CR) in this vulnerable population.
Design: We undertook a pilot randomised trial of CR following TAVI to inform the feasibility and design of a future randomised clinical trial (RCT).
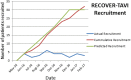
Participants: We screened patients undergoing TAVI at a single institution between June 2016 and February 2017.
Interventions: Participants were randomised post-TAVI to standard of care (control group) or standard of care plus exercise-based CR (intervention group).
Outcomes: We assessed recruitment and attrition rates, uptake of CR, and explored changes in 6-min walk test, Nottingham Activities of Daily Living, Fried and Edmonton Frailty scores and Hospital Anxiety and Depression Score, from baseline (30 days post TAVI) to 3 and 6 months post randomisation. We also undertook a parallel study to assess the use of the Kansas City Cardiomyopathy Questionnaire (KCCQ) in the post-TAVI population.
Results: Of 82 patients screened, 52 met the inclusion criteria and 27 were recruited (3 patients/month). In the intervention group, 10/13 (77%) completed the prescribed course of 6 sessions of CR (mean number of sessions attended 7.5, SD 4.25) over 6 weeks. At 6 months, all participants were retained for follow-up. There was apparent improvement in outcome scores at 3 and 6 months in control and CR groups. There were no recorded adverse events associated with the intervention of CR. The KCCQ was well accepted in 38 post-TAVI patients: mean summary score 72.6 (SD 22.6).
Conclusions: We have demonstrated the feasibility of recruiting post-TAVI patients into a randomised trial of CR. We will use the findings of this pilot trial to design a fully powered multicentre RCT to inform the provision of CR and support guideline development to optimise health-related quality of life outcomes in this vulnerable population. Retrospectively registered 3rd October 2016 clinicaltrials.gov NCT02921880.
Trial registration: Clinicaltrials.Gov identifier NCT02921880.
Keywords: Cardiac rehabilitation; Pilot study; Randomised controlled trial; Transcatheter aortic valve implantation.
Conflict of interest statement
MD has received research grants, consultancy and proctorship fees from Astra Zeneca, Eli Lilly, Abbott Vascular, Daiichi Sankyo, Daiichi Sankyo Lilly Alliance, Abbott Vascular, Sanofi, Medtronic, Boston Scientific, Edwards Lifesciences. NM has received honoraria, consultancy and proctorship fees from Abbott Vascular,, Medtronic, and Edwards Lifesciences. MS has received research grants, consultancy and proctorship fees from Medtronic, Edwards Lifesciences, St Jude (Now Abbott Vascular) and Boston Scientific. RST is the lead for the ongoing portfolio of Cochrane reviews of cardiac rehabilitation. RST is a named Scientific Advisors for ongoing the National Institute of. Health and Care Excellence (NICE) updated clinical guidelines for the management heart failure (CG108). HP is a member of the British Association for Cardiovascular Prevention and Rehabilitation (BACPR) and the Association of Chartered Physiotherapists in Cardiac Rehabilitation (ACPICR). HP Chaired the referenced ACPICR working group for the national standards document.Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous