Metformin modulates innate immune-mediated inflammation and early progression of NAFLD-associated hepatocellular carcinoma in zebrafish
- PMID: 30572006
- PMCID: PMC6436385
- DOI: 10.1016/j.jhep.2018.11.034
Metformin modulates innate immune-mediated inflammation and early progression of NAFLD-associated hepatocellular carcinoma in zebrafish
Abstract
Background & aims: Non-alcoholic fatty liver disease/non-alcoholic steatohepatitis (NAFLD/NASH) is an increasing clinical problem associated with progression to hepatocellular carcinoma (HCC). The effect of a high-fat diet on the early immune response in HCC is poorly understood, while the role of metformin in treating NAFLD and HCC remains controversial. Herein, we visualized the early immune responses in the liver and the effect of metformin on progression of HCC using optically transparent zebrafish.
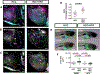
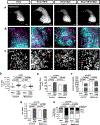
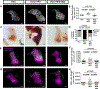
Methods: We used live imaging to visualize liver inflammation and disease progression in a NAFLD/NASH-HCC zebrafish model. We combined a high-fat diet with a transgenic zebrafish HCC model induced by hepatocyte-specific activated beta-catenin and assessed liver size, angiogenesis, micronuclei formation and inflammation in the liver. In addition, we probed the effects of metformin on immune cell composition and early HCC progression.
Results: We found that a high-fat diet induced an increase in liver size, enhanced angiogenesis, micronuclei formation and neutrophil infiltration in the liver. Although macrophage number was not affected by diet, a high-fat diet induced changes in macrophage morphology and polarization with an increase in liver associated TNFα-positive macrophages. Treatment with metformin altered macrophage polarization, reduced liver size and reduced micronuclei formation in NAFLD/NASH-associated HCC larvae. Moreover, a high-fat diet reduced T cell density in the liver, which was reversed by treatment with metformin.
Conclusions: These findings suggest that diet alters macrophage polarization and exacerbates the liver inflammatory microenvironment and cancer progression in a zebrafish model of NAFLD/NASH-associated HCC. Metformin specifically affects the progression induced by diet and modulates the immune response by affecting macrophage polarization and T cell infiltration, suggesting possible effects of metformin on tumor surveillance.
Lay summary: This paper reports a new zebrafish model that can be used to study the effects of diet on liver cancer. We found that a high-fat diet promotes non-resolving inflammation in the liver and enhances cancer progression. In addition, we found that metformin, a drug used to treat diabetes, inhibits high-fat diet-induced cancer progression in this model, by reducing diet-induced non-resolving inflammation and potentially restoring tumor surveillance.
Keywords: High-fat diet; Liver cancer; Metformin; NAFLD-associated HCC; NAFLD/NASH; Zebrafish model.
Copyright © 2018 European Association for the Study of the Liver. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

