Parkin inhibits BAK and BAX apoptotic function by distinct mechanisms during mitophagy
- PMID: 30573668
- PMCID: PMC6331729
- DOI: 10.15252/embj.201899916
Parkin inhibits BAK and BAX apoptotic function by distinct mechanisms during mitophagy
Abstract
The E3 ubiquitin ligase Parkin is a key effector of the removal of damaged mitochondria by mitophagy. Parkin determines cell fate in response to mitochondrial damage, with its loss promoting early onset Parkinson's disease and potentially also cancer progression. Controlling a cell's apoptotic response is essential to co-ordinate the removal of damaged mitochondria. We report that following mitochondrial damage-induced mitophagy, Parkin directly ubiquitinates the apoptotic effector protein BAK at a conserved lysine in its hydrophobic groove, a region that is crucial for BAK activation by BH3-only proteins and its homo-dimerisation during apoptosis. Ubiquitination inhibited BAK activity by impairing its activation and the formation of lethal BAK oligomers. Parkin also suppresses BAX-mediated apoptosis, but in the absence of BAX ubiquitination suggesting an indirect mechanism. In addition, we find that BAK-dependent mitochondrial outer membrane permeabilisation during apoptosis promotes PINK1-dependent Parkin activation. Hence, we propose that Parkin directly inhibits BAK to suppress errant apoptosis, thereby allowing the effective clearance of damaged mitochondria, but also promotes clearance of apoptotic mitochondria to limit their potential pro-inflammatory effect.
Keywords: BAK; BAX; Parkin; apoptosis; mitophagy.
© 2018 The Authors.
Figures

Schematic of potential effects of activated Parkin on the BCL‐2 family of apoptosis regulators.
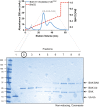
BAX/BAK‐deficient HeLa cells reconstituted with BAX and where indicated HA‐Parkin were treated with antimycin A and oligomycin (AO) for 2 h and then 1 μM of each of the BH3 mimetics ABT‐737 and S63485 for up to 6 h. Cell death was assessed at the indicated times by LDH assay. Data are mean ± SD of four independent experiments.
BAX/BAK‐deficient HeLa cells ectopically expressing BAX and HA‐Parkin were treated with antimycin A and oligomycin (AO, 3 h), MG132 (20 μM, 3.5 h) and BH3 mimetics (1 μM of each of ABT‐737 and S63485, 1 h). Cytosol and membrane fractions were immunoblotted as indicated. Graph shows densitometric analysis of non‐ubiquitinated membrane proteins (top, relative to untreated control) or cytochrome c release (bottom; cytosol/cytosol + membrane, relative to untreated controls) from three independent experiments. Error bars represent mean ± SD.
BAX/BAK‐deficient HeLa cells ectopically expressing BAX were treated and analysed as in (C).
UBA pull‐down of ubiquitinated proteins following antimycin A and oligomycin (AO) treatment of BAX/BAK‐deficient HeLa cells ectopically expressing HA‐Parkin and BAX for 3 h. Representative immunoblot from two independent experiments.

UBA pull‐down of ubiquitinated proteins following antimycin A and oligomycin (AO) treatment of wild‐type (WT) HeLa cells for 3 h. Representative immunoblot from two independent experiments.
Endogenous BAK is ubiquitinated in neuroblastoma cells upon mitochondrial damage. SH‐SY5Y cells ectopically expressing Parkin were treated with antimycin A and oligomycin (AO) for 2 h. Representative immunoblots of three independent experiments.
Endogenous BAK is ubiquitinated in MEFs upon mitochondrial damage. Wild‐type MEFs (WT) or Bak −/− Bax −/− MEFs expressing hBAK and ectopically expressing HA‐Parkin were treated with antimycin A and oligomycin (AO) with or without MG132 for 2 h. Lysates were immunoblotted as described. Note that endogenous mouse BAK was ubiquitinated and that ubiquitinated forms of mouse and human BAK (*) were reduced with MG132. Representative immunoblots of three independent experiments.
Timecourse of mitophagy induction. HeLa cells expressing HA‐Parkin were treated with antimycin A and oligomycin (AO) for the indicated times prior to immunoblotting of membrane fractions as described. Representative immunoblot from two independent experiments.
BAK −/− BAX −/− HeLa cells expressing BAK and HA‐Parkin were treated with antimycin A and oligomycin (AO) for 2 h prior to immunoprecipitation of BAK. Immunoprecipitates were incubated with the indicated ubiquitin linkage‐specific DUB prior to analysis of BAK by immunoblotting. Data representative of three independent experiments.

Subcellular fractionation of cytosol and membrane fractions of HeLa cells expressing HA‐Parkin following 2 h of antimycin A and oligomycin (AO) treatment. Representative immunoblot of three independent experiments.
Immunoblotting of whole cell lysates following treatment with antimycin A and oligomycin (AO) for 3 h with MG132 (20 μM). Graph shows densitometric analysis of non‐ubiquitinated proteins relative to untreated control from three independent experiments. Error bars represent mean ± SD.
UBA pull‐down of ubiquitinated proteins following antimycin A and oligomycin (AO) treatment for 3 h and treatment with the non‐specific DUB USP2 for 30 min at 37°C, representative immunoblot from two independent experiments.
Structure of human BAK (PDB 2IMS) with the hydrophobic binding groove comprising α‐helices 3–5 highlighted in blue. The ubiquitination site, K113, (red) resides in the binding groove. A modelled transmembrane anchor (α9) and the C‐terminal K210 localised to the inter‐membrane space are based on evidence that the BAK transmembrane anchor spans the mitochondrial outer membrane (Iyer et al, 2015). The orientation of the soluble portion (α‐helices 1–8) of BAK with respect to the mitochondrial outer membrane is hypothetical.
Whole cell lysates of HA‐Parkin and BAK variant‐expressing HeLa cells following 3 h antimycin A and oligomycin (AO) treatment with 30 min pre‐treatment with MG132 (20 μM), representative immunoblots from three independent experiments.

Lys113 at the hydrophobic groove of BAK is evolutionarily conserved.
Whole cell lysates of stable cell lines generated from BAK/BAX‐deficient HeLa re‐expressing BAK constructs and HA‐Parkin, immunoblotted as described.
Ubiquitination of BAK does not significantly alter its subcellular localisation. BAK −/− BAX −/− HeLa cells expressing HA‐Parkin together with wild‐type BAK or BAK K113R were treated with antimycin A and oligomycin (AO) for 2 h prior to subcellular fractionation into cytosol (C) and mitochondria‐enriched heavy membrane pellet (M) fractions. Fractions were immunoblotted as described. Representative immunoblot of two independent experiments.

HeLa cells with or without HA‐Parkin were treated with CCCP (10 μM), antimycin A and oligomycin (AO) for 2 h or GTPP (10 μM) for 6 h. Immunoblots are representative of three independent experiments. Graph shows densitometric analysis of non‐ubiquitinated proteins relative to untreated control from three independent experiments. Error bars represent mean ± SD.
SH‐SY5Y neuroblastoma cells expressing Cas9 together with a sgRNA targeting PRKN (sgPRKN) or a non‐targeting control (sgCon) were assessed for Parkin expression by immunoblotting of cytosolic fractions.
SH‐SY5Y cells generated in (B) were treated with antimycin A and oligomycin (AO) for 3 h prior to cell lysis and immunoblotting. Mono and di‐ubiquitinated BAK in Parkin‐expressing cells following AO are indicated (*). Data are representative of three independent experiments.
Domain architecture of human Parkin with positions of selected PD‐associated mutations. Ubl, ubiquitin‐like; IBR, in‐between‐RING (Gladkova et al, 2018). HeLa cells expressing wild‐type Parkin or the indicated PD‐associated Parkin mutants were treated with antimycin A and oligomycin (AO) for 2 h. Immunoblots are representative of three independent experiments. Graph shows densitometric analysis of non‐ubiquitinated proteins in AO‐treated samples relative to untreated control from three independent experiments. Error bars represent mean ± SD. All Parkin constructs were N‐terminally FLAG‐tagged except R104W.

BN–PAGE and SDS–PAGE of HeLa + HA‐Parkin cells treated with 1 μM of each of ABT‐737 and S63485 for 3 h (BH3), antimycin A and oligomycin (AO) for 2 h or a combination of the two. Experiment was performed in the presence of 10 μM QVD.oph and immunoblots are representative of three independent experiments.
UBA enrichment of ubiquitinated proteins from HeLa + HA‐Parkin cells, BAX −/− BAK −/− (DKO) HeLa cells or wild‐type (WT) HeLa cells expressing HA‐Parkin in response to 1 μM of each of ABT‐737 and S63485 for 3 h (BH3) in the presence of QVD.oph (10 μM). Representative of three independent experiments.
HeLa cells expressing HA‐Parkin and Cas9 were transduced with sgRNA targeting PINK1 or a non‐targeting sgRNA control, treated with AO (2 h) to stabilise PINK1 expression and immunoblotted for PINK1.
Parkin activity induced by apoptotic mitochondrial damage is PINK1‐dependent. UBA enrichment of ubiquitinated proteins from HeLa cells generated in (C) in response to 1 μM of each of ABT‐737 and S63485 for 3 h (BH3) in the presence of QVD.oph (10 μM). (*) ubiquitinated protein. Representative of two independent experiments.
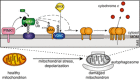
Schematic showing Parkin ubiquitination of BAK monomer in non‐apoptotic cells, but not the BAK homo‐dimer in cells undergoing apoptosis. In response to mitophagy stimuli, PINK1 phosphorylates ubiquitin to recruit Parkin, which in turn is phosphorylated by PINK1 to become activated to ubiquitinate monomeric BAK on K113. Mitochondrial outer membrane (MOM) permeabilisation driven by BAK oligomers during apoptosis provokes Parkin activity in a PINK1‐dependent manner. Although Parkin can ubiquitinate mitochondrial outer membrane substrates (e.g. Mfn2) to promote mitochondrial clearance, it cannot ubiquitinate dimerised BAK on K113.

HeLa cells were treated with antimycin A and oligomycin (AO) for 2 h or with 1 μM of each of ABT‐737 and S63485 for 3 h (BH3) in the presence of QVD.oph (10 μM). Representative of three independent experiments.
HeLa cells engineered in Fig 4C were treated with 1 μM of each of ABT‐737 and S63485 for 3 h (BH3) in the presence of QVD.oph (10 μM) prior to subcellular fractionation of cytosol (C) and mitochondria‐enriched heavy membrane (M) fractions. Representative of two independent experiments.

Schematic of fluorescence‐based liposome assay to measure BAK activation on model membranes.
Non‐reducing SDS–PAGE and immunoblot of recombinant BAK and BAK‐Ub following treatment with 25 mM TCEP.
Timecourse monitoring fluorescence increase on liposomes following addition of 4 μM BID‐BH3 peptide to 50 nM BAK or BAK‐Ub in the presence or absence of 25 mM TCEP. Data are mean ± SD of three independent experiments.
BN–PAGE of BAK and BAK‐Ub on liposomes following incubation with 4 μM BID‐BH3 peptide in the presence or absence of 25 mM TCEP, immunoblotted for BAK. Data representative of three independent experiments. Graphs shows densitometric analysis of the BAK tetramer (4×) from three or two independent experiments. Error bars represent mean ± SD for n = 3 or mean and range for n = 2.

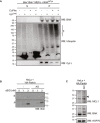
Immunoblot of whole cell lysates of MEFs stably expressing ΔCys variants of BAK.
Conjugation of ubiquitin to BAK K113C impairs BAK apoptotic function. Mitochondrially enriched membranes from MEFs expressing BAK or BAK K113C were cross‐linked (CuPhe) with UbG76C and treated with cBID for 30 min at 30°C. Supernatant (S) and pellet (P) fractions were then immunoblotted for BAK under non‐reducing conditions, or cytochrome c or ubiquitin under reducing conditions. Representative of three independent experiments. Note that the incorporation of recombinant UbG76C on higher molecular weight proteins is likely due to incomplete reduction on reducing SDS–PAGE.
Parkin‐mediated ubiquitination limits BAK activation. HeLa + HA‐Parkin cells were treated with antimycin A and oligomycin (AO) for 2 h prior to induction of apoptosis with 1 μM of each of the BH3 mimetics ABT‐737 and S63485 (BH3 mim) for 1 h. Intracellular flow cytometry was performed using the conformation‐specific anti‐BAK antibody G317‐2. Representative histograms are shown and the fold increase in mean fluorescence intensity (MFI) is plotted from three independent experiments with error bars showing SD. Data are normalised to untreated control.
Parkin‐mediated ubiquitination limits BAK apoptotic activity. BAK −/− BAX −/− HeLa cells expressing HA‐Parkin and BAK were treated with AO (2 h) prior to isolation of mitochondria and treatment with recombinant cBID and analysis of cytochrome c release. Immunoblots are representative of three independent experiments. Graph shows densitometric analysis of three independent experiments of cytochrome c in the supernatant fraction. Error bars represent mean ± SD.
Parkin activity limits cytochrome c release. HeLa cells or HeLa cells expressing Parkin were treated and analysed as in (D). Immunoblots are representative of three independent experiments. Ubiquitinated BAK indicated (arrow). Graph shows densitometric analysis of three independent experiments showing cytochrome c release (supernatant/supernatant + membrane) relative to 100 nM cBID as 100% release. Error bars represent mean ± SD.
- A
Mitochondria‐enriched membrane fraction from Bax −/− Bak −/− MEFs expressing BAK∆Cys were incubated with UbG76C (100 μM) in the presence or absence of oxidant copper phenanthroline (CuPhe, 1 mM) for 30 min prior to separation of supernatant and membrane fractions and immunoblotting as indicated. Note the incorporation of exogenous UbG76C into high molecular weight species (*) in the membrane fraction following treatment with oxidant. (**), UbG76C disulphide‐linked dimer.
- B
HeLa cells expressing HA‐Parkin were treated with antimycin and oligomycin (AO) for 2 h prior to fractionation into cytosol (C) and heavy membrane (M). Membrane fractions were then incubated with cBID as indicated. Incorporated cBID was then determined by centrifugation to separate the supernatant (S) from the membrane pellet (P) prior to immunoblotting.
- C
HeLa + HA‐Parkin cells were treated with antimycin and oligomycin (AO) for 2 h prior to harvesting. Samples were immunoblotted as described. Ubiquitinated MCL1 (arrowhead).
References
-
- Alsop AE, Fennell SC, Bartolo RC, Tan IK, Dewson G, Kluck RM (2015) Dissociation of Bak alpha1 helix from the core and latch domains is required for apoptosis. Nat Commun 6: 6841 - PubMed
-
- Arkinson C, Walden H (2018) Parkin function in Parkinson's disease. Science 360: 267–268 - PubMed
-
- Bernardini JP, Lazarou M, Dewson G (2017) Parkin and mitophagy in cancer. Oncogene 36: 1315–1327 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

