Survival of Hepatocellular Carcinoma Patients Treated with Sorafenib beyond Progression
- PMID: 30574480
- PMCID: PMC6288626
- DOI: 10.1159/000487635
Survival of Hepatocellular Carcinoma Patients Treated with Sorafenib beyond Progression
Abstract
Background/aim: Sorafenib leads to improved survival in advanced hepatocellular carcinoma (HCC) patients. Continuation of sorafenib beyond progression has been a possible treatment strategy when further approved therapeutic agents are lacking.
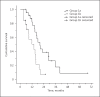
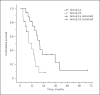
Methods: We performed a retrospective analysis of all HCC patients at our institution with documented disease progression under treatment with sorafenib. Overall survival (OS) from start of sorafenib treatment was compared between patients who received sorafenib for > 3 weeks beyond progression (group 1) and those who discontinued sorafenib ≤3 weeks after progression (group 2). Group 1 was further subdivided into those patients who received sorafenib for > 3 months (group 1a) and those who received it for ≤3 months (group 1b).
Results: A total of 71 patients were analyzed. Median OS for all patients was 15.4 months. OS in group 1 (15.6 months) and 2 (13.0 months) was similar (p = 0.90). Patients in group 1a showed significantly prolonged median OS (19.7 months) compared to that of patients in group 1b (13.6 months, p = 0.004), and they showed a trend towards prolonged OS compared to group 2 (p = 0.126). For patients with a poor prognosis according to their Child-Pugh stage, performance status, alpha-fetoprotein, and response to prior sorafenib treatment, OS was significantly prolonged in group 1 versus group 2 (12.1 vs. 6.4 months, p = 0.019).
Conclusion: In HCC patients, continuing sorafenib beyond progression for > 3 months is associated with improved survival compared to discontinuing sorafenib within 3 months. Furthermore, patients with a poor prognosis who continue sorafenib beyond progression in general show significantly prolonged survival.
Keywords: Hepatocellular carcinoma; Molecular-targeted therapies; Progression; Sorafenib; Survival; Therapeutics; Therapy; Treatment.
Figures


References
-
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. - PubMed
-
- Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. - PubMed
-
- Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, Liu J, Wang J, Tak WY, Pan H, Burock K, Zou J, Voliotis D, Guan Z. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34. - PubMed
-
- Reig M, Rimola J, Torres F, Darnell A, Rodriguez-Lope C, Forner A, Llarch N, Rios J, Ayuso C, Bruix J. Postprogression survival of patients with advanced hepatocellular carcinoma: rationale for second-line trial design. Hepatology. 2013;58:2023–2031. - PubMed
-
- Miyahara K, Nouso K, Morimoto Y, Takeuchi Y, Hagihara H, Kuwaki K, Onishi H, Ikeda F, Miyake Y, Nakamura S, Shiraha H, Takaki A, Iwadou S, Kobayashi Y, Takaguchi K, Takuma Y, Takabatake H, Sakaguchi K, Yamamoto K, Okayama Liver Cancer Group Efficacy of sorafenib beyond first progression in patients with metastatic hepatocellular carcinoma. Hepatol Res. 2014;44:296–301. - PubMed
LinkOut - more resources
Full Text Sources

