Vaporized E-Cigarette Liquids Induce Ion Transport Dysfunction in Airway Epithelia
- PMID: 30576219
- PMCID: PMC6670031
- DOI: 10.1165/rcmb.2017-0432OC
Vaporized E-Cigarette Liquids Induce Ion Transport Dysfunction in Airway Epithelia
Abstract
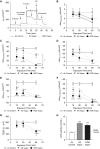
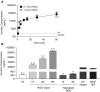
Cigarette smoking is associated with chronic obstructive pulmonary disease and chronic bronchitis. Acquired ion transport abnormalities, including cystic fibrosis transmembrane conductance regulator (CFTR) dysfunction, caused by cigarette smoking have been proposed as potential mechanisms for mucus obstruction in chronic bronchitis. Although e-cigarette use is popular and perceived to be safe, whether it harms the airways via mechanisms altering ion transport remains unclear. In the present study, we sought to determine if e-cigarette vapor, like cigarette smoke, has the potential to induce acquired CFTR dysfunction, and to what degree. Electrophysiological methods demonstrated reduced chloride transport caused by vaporized e-cigarette liquid or vegetable glycerin at various exposures (30 min, 57.2% and 14.4% respectively, vs. control; P < 0.0001), but not by unvaporized liquid (60 min, 17.6% vs. untreated), indicating that thermal degradation of these products is required to induce the observed defects. We also observed reduced ATP-dependent responses (-10.8 ± 3.0 vs. -18.8 ± 5.1 μA/cm2 control) and epithelial sodium channel activity (95.8% reduction) in primary human bronchial epithelial cells after 5 minutes, suggesting that exposures dramatically inhibit epithelial ion transport beyond CFTR, even without diminished transepithelial resistance or cytotoxicity. Vaporizing e-cigarette liquid produced reactive aldehydes, including acrolein (shown to induce acquired CFTR dysfunction), as quantified by mass spectrometry, demonstrating that respiratory toxicants in cigarette smoke can also be found in e-cigarette vapor (30 min air, 224.5 ± 15.99; unvaporized liquid, 284.8 ± 35.03; vapor, 54,468 ± 3,908 ng/ml; P < 0.0001). E-cigarettes can induce ion channel dysfunction in airway epithelial cells, partly through acrolein production. These findings indicate a heretofore unknown toxicity of e-cigarette use known to be associated with chronic bronchitis onset and progression, as well as with chronic obstructive pulmonary disease severity.
Keywords: acquired cystic fibrosis transmembrane conductance regulator dysfunction; chronic obstructive pulmonary disease; e-cigarette; ion transport.
Figures





Comment in
-
Vaping Away Epithelial Integrity.Am J Respir Cell Mol Biol. 2019 Aug;61(2):127-129. doi: 10.1165/rcmb.2019-0016ED. Am J Respir Cell Mol Biol. 2019. PMID: 30726103 Free PMC article. No abstract available.
References
-
- Mannino DM, Buist AS. Global burden of COPD: risk factors, prevalence, and future trends. Lancet. 2007;370:765–773. - PubMed
-
- Rowe SM, Miller S, Sorscher EJ. Cystic fibrosis. N Engl J Med. 2005;352:1992–2001. - PubMed
-
- Kreindler JL, Jackson AD, Kemp PA, Bridges RJ, Danahay H. Inhibition of chloride secretion in human bronchial epithelial cells by cigarette smoke extract. Am J Physiol Lung Cell Mol Physiol. 2005;288:L894–L902. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

