Glioblastoma: Microenvironment and Niche Concept
- PMID: 30577488
- PMCID: PMC6357107
- DOI: 10.3390/cancers11010005
Glioblastoma: Microenvironment and Niche Concept
Abstract
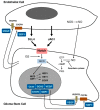
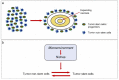
The niche concept was originally developed to describe the location of normal neural stem cells (NSCs) in the subependymal layer of the sub-ventricular zone. In this paper, its significance has been extended to the location of tumor stem cells in glioblastoma (GB) to discuss the relationship between GB stem cells (GSCs) and endothelial cells (ECs). Their interaction is basically conceived as responsible for tumor growth, invasion and recurrence. Niches are described as the points of utmost expression of the tumor microenvironment (TME), therefore including everything in the tumor except for tumor cells: NSCs, reactive astrocytes, ECs, glioma-associated microglia/macrophages (GAMs), myeloid cells, pericytes, fibroblasts, etc. and all intrinsic and extrinsic signaling pathways. Perivascular (PVNs), perinecrotic (PNNs) and invasive niches were described from the pathological point of view, highlighting the basic significance of the EC/tumor stem cell couple. PNN development was reinterpreted based on the concept that hyperproliferative areas of GB are composed of GSCs/progenitors. TME was depicted in its function as the main regulator of everything that happens in the tumor. A particular emphasis was given to GAMs, pericytes and reactive astrocytes as important elements affecting proliferation, growth, invasion and resistance to therapies of tumor cells.
Keywords: glioblastoma; microenvironment; niche; pericytes; reactive astrocytes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Schiffer D., Mellai M., Annovazzi L., Casalone C., Cassoni P. Tumor Microenvironment: Perivascular and Perinecrotic Niches in Gliomas. In: Morgan L.R., editor. Molecular Considerations and Evolving Surgical Management Issues in the Treatment of Patients with a Brain Tumor. 1st ed. InTech; Rjieka, Croatia: 2015. pp. 49–82.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

