Tuning recombinant protein expression to match secretion capacity
- PMID: 30577801
- PMCID: PMC6303999
- DOI: 10.1186/s12934-018-1047-z
Tuning recombinant protein expression to match secretion capacity
Abstract
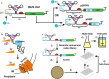
Background: The secretion of recombinant disulfide-bond containing proteins into the periplasm of Gram-negative bacterial hosts, such as E. coli, has many advantages that can facilitate product isolation, quality and activity. However, the secretion machinery of E. coli has a limited capacity and can become overloaded, leading to cytoplasmic retention of product; which can negatively impact cell viability and biomass accumulation. Fine control over recombinant gene expression offers the potential to avoid this overload by matching expression levels to the host secretion capacity.
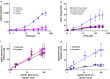
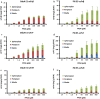
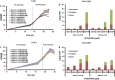
Results: Here we report the application of the RiboTite gene expression control system to achieve this by finely controlling cellular expression levels. The level of control afforded by this system allows cell viability to be maintained, permitting production of high-quality, active product with enhanced volumetric titres.
Conclusions: The methods and systems reported expand the tools available for the production of disulfide-bond containing proteins, including antibody fragments, in bacterial hosts.
Keywords: Antibody fragments; Codon usage; Periplasmic secretion; Riboswitches; Signal peptides.
Figures

References
MeSH terms
Substances
Grants and funding
- EP/I033270/1/Engineering and Physical Sciences Research Council
- BB/M011259/1/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/M011208/1/Biotechnology and Biological Sciences Research Council/United Kingdom
- BB/K014773/1/Biotechnology and Biological Sciences Research Council/United Kingdom
- 131836/Innovate UK
LinkOut - more resources
Full Text Sources
Other Literature Sources

