Epigenome-wide meta-analysis of DNA methylation and childhood asthma
- PMID: 30579849
- PMCID: PMC6556405
- DOI: 10.1016/j.jaci.2018.11.043
Epigenome-wide meta-analysis of DNA methylation and childhood asthma
Abstract
Background: Epigenetic mechanisms, including methylation, can contribute to childhood asthma. Identifying DNA methylation profiles in asthmatic patients can inform disease pathogenesis.
Objective: We sought to identify differential DNA methylation in newborns and children related to childhood asthma.
Methods: Within the Pregnancy And Childhood Epigenetics consortium, we performed epigenome-wide meta-analyses of school-age asthma in relation to CpG methylation (Illumina450K) in blood measured either in newborns, in prospective analyses, or cross-sectionally in school-aged children. We also identified differentially methylated regions.
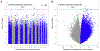
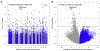
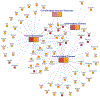
Results: In newborns (8 cohorts, 668 cases), 9 CpGs (and 35 regions) were differentially methylated (epigenome-wide significance, false discovery rate < 0.05) in relation to asthma development. In a cross-sectional meta-analysis of asthma and methylation in children (9 cohorts, 631 cases), we identified 179 CpGs (false discovery rate < 0.05) and 36 differentially methylated regions. In replication studies of methylation in other tissues, most of the 179 CpGs discovered in blood replicated, despite smaller sample sizes, in studies of nasal respiratory epithelium or eosinophils. Pathway analyses highlighted enrichment for asthma-relevant immune processes and overlap in pathways enriched both in newborns and children. Gene expression correlated with methylation at most loci. Functional annotation supports a regulatory effect on gene expression at many asthma-associated CpGs. Several implicated genes are targets for approved or experimental drugs, including IL5RA and KCNH2.
Conclusion: Novel loci differentially methylated in newborns represent potential biomarkers of risk of asthma by school age. Cross-sectional associations in children can reflect both risk for and effects of disease. Asthma-related differential methylation in blood in children was substantially replicated in eosinophils and respiratory epithelium.
Keywords: Epigenetics; asthma; childhood; drug development; methylation; newborn.
Published by Elsevier Inc.
Conflict of interest statement
Disclosure of potential conflict of interest: C. Ruiz-Arenas receives grant support from Agència de Gestió d’Ajuts Universitaris i de Recerca. S. S. Oh, C. Eng, and E. G. Burchard receive grant support from the NIH and the Tobacco-Related Disease Research Program. I. V. Yang and C. V. Breton receive grant support from the National Institutes of Health (NIH). C. Söderhäll receives grant support from several competitive grants from public and private sources and receives royalties from book chapters in study material. R. Arathimos and G. C. Sharp receive support from the Medical Research Council. E. Kajantie receives grant support from the European Commission, Academy of Finland, Foundation for Pediatric Research, Sigrid Jus×lius Foundation, Signe and Ane Gyllenberg Foundation, and Juho Vainio Foundation. G. Pershagen receives grant support from the Swedish Research Council. C. L. Relton receives grant support from Wellcome Trust. C. Almqvist receives grant support from the Swedish Research Council through the Swedish Initiative for Research on Microdata in the Social And Medical Sciences (SIMSAM) framework, Stockholm County Council (ALF-projects), Swedish Heart-Lung Foundation, and Swedish Asthma and Allergy Association’s Research Foundation. A. J. Henderson receives grant support from the Medical Research Council and Wellcome Trust. E. Melén received grant support from the European Research Council during conduct of the study and lecture fees from Thermo Fisher Scientific and Meda outside the submitted work. G. H. Koppelman receives grant support from the Lung Foundation of the Netherlands, MEDALL EU FP7, the UBBO EMMIUS Foundation, TEVA The Netherlands, Vertex, GlaxoSmithKline, and the TETRI Foundation. The rest of the authors declare that they have no relevant conflicts of interest.
Figures
References
-
- Bisgaard H, Szefler S. Prevalence of asthma-like symptoms in young children. Pediatr Pulmonol 2007;42:723–8. - PubMed
-
- Wjst M, Sargurupremraj M, Arnold M. Genome-wide association studies in asthma: what they really told us about pathogenesis. Curr Opin Allergy Clin Immunol 2013;13:112–8. - PubMed
-
- Weiss ST, Silverman EK. Pro: genome-wide association studies (GWAS) in asthma. Am J Respir Crit Care Med 2011;184:631–3. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- G0401540/MRC_/Medical Research Council/United Kingdom
- Wellcome Trust./International
- ZIA ES025045/ImNIH/Intramural NIH HHS/United States
- ZIA ES049019/ImNIH/Intramural NIH HHS/United States
- T32 HL007085/HL/NHLBI NIH HHS/United States
- R01 HL097163/HL/NHLBI NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
- R01 HL132321/HL/NHLBI NIH HHS/United States
- R33 HL120770/HL/NHLBI NIH HHS/United States
- MC_UU_00011/5/MRC_/Medical Research Council/United Kingdom
- G9815508/MRC_/Medical Research Council/United Kingdom
- R01 AI091905/AI/NIAID NIH HHS/United States
- MC_UU_12013/2/MRC_/Medical Research Council/United Kingdom

