Role of zinc insufficiency in fetal alveolar macrophage dysfunction and RSV exacerbation associated with fetal ethanol exposure
- PMID: 30580016
- PMCID: PMC6586529
- DOI: 10.1016/j.alcohol.2018.11.007
Role of zinc insufficiency in fetal alveolar macrophage dysfunction and RSV exacerbation associated with fetal ethanol exposure
Abstract
Background: We previously reported that maternal alcohol use significantly increases the risk of sepsis in premature and term newborns. In the mouse, fetal ethanol exposure results in an immunosuppressed phenotype for the alveolar macrophage (AM) and decreases bacterial phagocytosis. In pregnant mice, ethanol decreased AM zinc homeostasis, which contributed to immunosuppression and impaired AM phagocytosis. In this study, we explored whether ethanol-induced zinc insufficiency extended to the pup AMs and contributed to immunosuppression and exacerbated viral lung infections.
Methods: C57BL/6 female mice were fed a liquid diet with 25% ethanol-derived calories or pair-fed a control diet with 25% of calories as maltose-dextrin. Some pup AMs were treated in vitro with zinc acetate before measuring zinc pools or transporter expression and bacteria phagocytosis. Some dams were fed additional zinc supplements in the ethanol or control diets, and then we assessed pup AM zinc pools, zinc transporters, and the immunosuppressant TGFβ1. On postnatal day 10, some pups were given intranasal saline or respiratory syncytial virus (RSV), and then AM RSV phagocytosis and the RSV burden in the airway lining fluid were assessed.
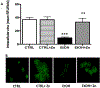
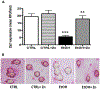
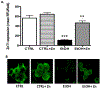
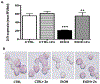
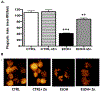
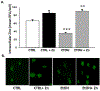
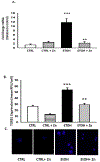
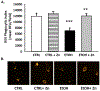
Results: Fetal ethanol exposure decreased pup AM zinc pools, zinc transporter expression, and bacterial clearance, but in vitro zinc treatments reversed these alterations. In addition, the expected ethanol-induced increase in TGFβ1 and immunosuppression were associated with decreased RSV phagocytosis and exacerbated RSV infections. However, additional maternal zinc supplements blocked the ethanol-induced perturbations in the pup AM zinc homeostasis and TGFβ1 immunosuppression, thereby improving RSV phagocytosis and attenuating the RSV burden in the lung.
Conclusion: These studies suggest that, despite normal maternal dietary zinc intake, in utero alcohol exposure results in zinc insufficiency, which contributes to compromised neonatal AM immune functions, thereby increasing the risk of bacterial and viral infections.
Keywords: Alveolar macrophage; Fetal ethanol exposure; Phagocytic function; Zinc insufficiency; Zinc transporters.
Copyright © 2018 Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Liposomal Glutathione Augments Immune Defenses against Respiratory Syncytial Virus in Neonatal Mice Exposed in Utero to Ethanol.Antioxidants (Basel). 2024 Jan 23;13(2):137. doi: 10.3390/antiox13020137. Antioxidants (Basel). 2024. PMID: 38397736 Free PMC article.
-
Sexual dimorphism in lung transcriptomic adaptations in fetal alcohol spectrum disorders.Respir Res. 2025 Jan 8;26(1):6. doi: 10.1186/s12931-025-03094-z. Respir Res. 2025. PMID: 39780208 Free PMC article.
-
Respiratory syncytial virus vaccination during pregnancy for improving infant outcomes.Cochrane Database Syst Rev. 2024 May 2;5(5):CD015134. doi: 10.1002/14651858.CD015134.pub2. Cochrane Database Syst Rev. 2024. PMID: 38695784 Free PMC article.
-
Calcium supplementation commencing before or early in pregnancy, or food fortification with calcium, for preventing hypertensive disorders of pregnancy.Cochrane Database Syst Rev. 2017 Sep 26;9(9):CD011192. doi: 10.1002/14651858.CD011192.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2019 Sep 16;9:CD011192. doi: 10.1002/14651858.CD011192.pub3. PMID: 28949421 Free PMC article. Updated.
-
Ethanol for preventing preterm birth in threatened preterm labor.Cochrane Database Syst Rev. 2015 Nov 5;2015(11):CD011445. doi: 10.1002/14651858.CD011445.pub2. Cochrane Database Syst Rev. 2015. PMID: 26544539 Free PMC article.
Cited by
-
Liposomal Glutathione Augments Immune Defenses against Respiratory Syncytial Virus in Neonatal Mice Exposed in Utero to Ethanol.Antioxidants (Basel). 2024 Jan 23;13(2):137. doi: 10.3390/antiox13020137. Antioxidants (Basel). 2024. PMID: 38397736 Free PMC article.
-
Dual therapy with zinc acetate and rifaximin prevents from ethanol-induced liver fibrosis by maintaining intestinal barrier integrity.World J Gastroenterol. 2021 Dec 28;27(48):8323-8342. doi: 10.3748/wjg.v27.i48.8323. World J Gastroenterol. 2021. PMID: 35068872 Free PMC article.
-
Zinc and respiratory tract infections: Perspectives for COVID‑19 (Review).Int J Mol Med. 2020 Jul;46(1):17-26. doi: 10.3892/ijmm.2020.4575. Epub 2020 Apr 14. Int J Mol Med. 2020. PMID: 32319538 Free PMC article. Review.
-
Can food and food supplements be deployed in the fight against the COVID 19 pandemic?Biochim Biophys Acta Gen Subj. 2021 Feb;1865(2):129801. doi: 10.1016/j.bbagen.2020.129801. Epub 2020 Nov 22. Biochim Biophys Acta Gen Subj. 2021. PMID: 33238195 Free PMC article. Review.
-
Role of different types of nanomaterials against diagnosis, prevention and therapy of COVID-19.Sustain Cities Soc. 2021 Sep;72:103046. doi: 10.1016/j.scs.2021.103046. Epub 2021 May 25. Sustain Cities Soc. 2021. PMID: 34055576 Free PMC article. Review.
References
-
- Akison LK, Kuo J, Reid N, Boyd RN, & Moritz KM (2018). Effect of Choline Supplementation on Neurological, Cognitive, and Behavioral Outcomes in Offspring Arising from Alcohol Exposure During Development: A Quantitative Systematic Review of Clinical and Preclinical Studies. Alcohol Clin Exp Res, 42(9), 1591–1611. doi:10.1111/acer.13817 - DOI - PubMed
-
- Beach RS, Gershwin ME, & Hurley LS (1982a). Gestational zinc deprivation in mice: persistence of immunodeficiency for three generations. Science, 218(4571), 469–471. - PubMed
-
- Beach RS, Gershwin ME, & Hurley LS (1982b). Reversibility of development retardation following murine fetal zinc deprivation. J Nutr, 112(6), 1169–1181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

