Hippo Deficiency Leads to Cardiac Dysfunction Accompanied by Cardiomyocyte Dedifferentiation During Pressure Overload
- PMID: 30582455
- PMCID: PMC6645688
- DOI: 10.1161/CIRCRESAHA.118.314048
Hippo Deficiency Leads to Cardiac Dysfunction Accompanied by Cardiomyocyte Dedifferentiation During Pressure Overload
Abstract
Rationale: The Hippo pathway plays an important role in determining organ size through regulation of cell proliferation and apoptosis. Hippo inactivation and consequent activation of YAP (Yes-associated protein), a transcription cofactor, have been proposed as a strategy to promote myocardial regeneration after myocardial infarction. However, the long-term effects of Hippo deficiency on cardiac function under stress remain unknown.
Objective: We investigated the long-term effect of Hippo deficiency on cardiac function in the presence of pressure overload (PO).
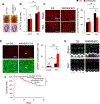
Methods and results: We used mice with cardiac-specific homozygous knockout of WW45 (WW45cKO), in which activation of Mst1 (Mammalian sterile 20-like 1) and Lats2 (large tumor suppressor kinase 2), the upstream kinases of the Hippo pathway, is effectively suppressed because of the absence of the scaffolding protein. We used male mice at 3 to 4 month of age in all animal experiments. We subjected WW45cKO mice to transverse aortic constriction for up to 12 weeks. WW45cKO mice exhibited higher levels of nuclear YAP in cardiomyocytes during PO. Unexpectedly, the progression of cardiac dysfunction induced by PO was exacerbated in WW45cKO mice, despite decreased apoptosis and activated cardiomyocyte cell cycle reentry. WW45cKO mice exhibited cardiomyocyte sarcomere disarray and upregulation of TEAD1 (transcriptional enhancer factor) target genes involved in cardiomyocyte dedifferentiation during PO. Genetic and pharmacological inactivation of the YAP-TEAD1 pathway reduced the PO-induced cardiac dysfunction in WW45cKO mice and attenuated cardiomyocyte dedifferentiation. Furthermore, the YAP-TEAD1 pathway upregulated OSM (oncostatin M) and OSM receptors, which played an essential role in mediating cardiomyocyte dedifferentiation. OSM also upregulated YAP and TEAD1 and promoted cardiomyocyte dedifferentiation, indicating the existence of a positive feedback mechanism consisting of YAP, TEAD1, and OSM.
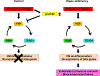
Conclusions: Although activation of YAP promotes cardiomyocyte regeneration after cardiac injury, it induces cardiomyocyte dedifferentiation and heart failure in the long-term in the presence of PO through activation of the YAP-TEAD1-OSM positive feedback mechanism.
Keywords: apoptosis; cell cycle; cell proliferation; heart failure; mice.
Conflict of interest statement
The authors have declared that no conflict of interest exists.
Figures





References
-
- Ikeda S and Sadoshima J. Regulation of Myocardial Cell Growth and Death by the Hippo Pathway. Circulation journal : official journal of the Japanese Circulation Society 2016. - PubMed
-
- Yamamoto S, Yang G, Zablocki D, Liu J, Hong C, Kim SJ, Soler S, Odashima M, Thaisz J, Yehia G, Molina CA, Yatani A, Vatner DE, Vatner SF and Sadoshima J. Activation of Mst1 causes dilated cardiomyopathy by stimulating apoptosis without compensatory ventricular myocyte hypertrophy. The Journal of clinical investigation 2003;111:1463–74. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL102738/HL/NHLBI NIH HHS/United States
- R01 HL091469/HL/NHLBI NIH HHS/United States
- R01 HL071894/HL/NHLBI NIH HHS/United States
- K02 HL114749/HL/NHLBI NIH HHS/United States
- R01 HL138720/HL/NHLBI NIH HHS/United States
- R01 HL130356/HL/NHLBI NIH HHS/United States
- R21 HL098802/HL/NHLBI NIH HHS/United States
- R01 HL067724/HL/NHLBI NIH HHS/United States
- R01 HL105826/HL/NHLBI NIH HHS/United States
- R01 AG023039/AG/NIA NIH HHS/United States
- R01 HL127339/HL/NHLBI NIH HHS/United States
- R01 HL112330/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

