Degeneration of dopaminergic circuitry influences depressive symptoms in Lewy body disorders
- PMID: 30582885
- PMCID: PMC6767514
- DOI: 10.1111/bpa.12697
Degeneration of dopaminergic circuitry influences depressive symptoms in Lewy body disorders
Abstract
Aims: Depression is commonly observed even in prodromal stages of Lewy body disorders (LBD), and is associated with cognitive impairment and a faster rate of cognitive decline. Given the role of dopamine in the development of movement disorders, but also in motivation and reward, we investigated neurodegenerative pathology in dopaminergic circuitry in Parkinson's disease (PD), PD with dementia (PDD) and dementia with Lewy bodies (DLB) patients in relation to depressive symptoms.
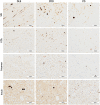
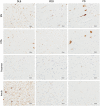
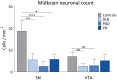
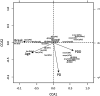
Methods: α-synuclein, hyperphosphorylated tau and amyloid-beta pathology was assessed in 17 DLB, 14 PDD and 8 PD cases within striatal and midbrain subregions, with neuronal cell density assessed in substantia nigra and ventral tegmental area. Additionally, we used a structural equation modeling (SEM) approach to investigate the extent to which brain connectivity might influence the deposition of pathological proteins within dopaminergic pathways.
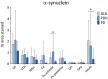
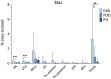
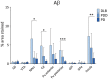
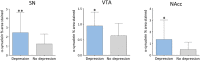
Results: A significantly higher α-synuclein burden was observed in the substantia nigra (P = 0.006), ventral tegmental area (P = 0.011) and nucleus accumbens (P = 0.031) in LBD patients with depression. Significant negative correlations were observed between cell density in substantia nigra with Lewy body (LB) Braak stage (P = 0.013), whereas cell density in ventral tegmental area showed negative correlations with LB Braak stage (P = 0.026) and neurofibrillary tangle Braak stage (P = 0.007).
Conclusions: Dopaminergic α-synuclein pathology appears to drive depression. Selective targeting of dopaminergic pathways may therefore provide symptomatic relief for depressive symptoms in LBD patients.
Keywords: dementia with Lewy bodies; depression; dopaminergic pathways; α-synuclein.
© 2018 The Authors Brain Pathology published by John Wiley & Sons Ltd on behalf of International Society of Neuropathology.
Figures

References
-
- Aarsland D, Ballard C, Larsen JP, McKeith I (2001) A comparative study of psychiatric symptoms in dementia with Lewy bodies and Parkinson's disease with and without dementia. Int J Geriatr Psych 16:528–536. - PubMed
-
- Alexander GE, Crutcher MD, DeLong MR (1990) Basal ganglia‐thalamocortical circuits: parallel substrates for motor, oculomotor, “prefrontal” and “limbic” functions. Prog Brain Res 85:119–146. - PubMed
-
- Alexopoulos GS, Abrams RC, Young RC, Shamoian CA (1988) Cornell scale for depression in Dementia. Biol Psychiatry 23:271–284. - PubMed

