Contribution of endogenous glucagon-like peptide-1 to changes in glucose metabolism and islet function in people with type 2 diabetes four weeks after Roux-en-Y gastric bypass (RYGB)
- PMID: 30586575
- PMCID: PMC6401231
- DOI: 10.1016/j.metabol.2018.12.005
Contribution of endogenous glucagon-like peptide-1 to changes in glucose metabolism and islet function in people with type 2 diabetes four weeks after Roux-en-Y gastric bypass (RYGB)
Abstract
Glucagon-Like Peptide-1 (GLP-1) is an insulin secretagogue which is elevated after Roux-en-Y Gastric Bypass (RYGB). However, its contribution to glucose metabolism after RYGB remains uncertain.
Aims: We tested the hypothesis that GLP-1 lowers postprandial glucose concentrations and improves β-cell function after RYGB.
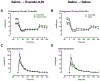
Materials and methods: To address these questions we used a labeled mixed meal to assess glucose metabolism and islet function in 12 obese subjects with type 2 diabetes studied before and four weeks after RYGB. During the post-RYGB study subjects were randomly assigned to receive an infusion of either saline or Exendin-9,39 a competitive antagonist of GLP-1 at its receptor. Exendin-9,39 was infused at 300 pmol/kg/min for 6 h. All subjects underwent RYGB for medically-complicated obesity.
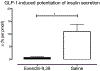
Results: Exendin-9,39 resulted in increased integrated incremental postprandial glucose concentrations (181 ± 154 vs. 582 ± 129 mmol per 6 h, p = 0.02). In contrast, this was unchanged in the presence of saline (275 ± 88 vs. 315 ± 66 mmol per 6 h, p = 0.56) after RYGB. Exendin-9,39 also impaired β-cell responsivity to glucose but did not alter Disposition Index (DI).
Conclusions: These data indicate that the elevated GLP-1 concentrations that occur early after RYGB improve postprandial glucose tolerance by enhancing postprandial insulin secretion.
Copyright © 2018 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures
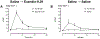


References
-
- Buchwald H, Avidor Y, Braunwald E, Jensen MD, Pories W, Fahrbach K, et al. Bariatric surgery: a systematic review and meta-analysis. Jama. 2004;292:1724–37. - PubMed
-
- Kashyap SR, Bhatt DL, Wolski K, Watanabe RM, Abdul-Ghani M, Abood B, et al. Metabolic Effects of Bariatric Surgery in Patients With Moderate Obesity and Type 2 Diabetes: Analysis of a randomized control trial comparing surgery with intensive medical treatment. Diabetes Care. 2013;36:2175–82. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

