Phase II Study of Weekly Amrubicin for Refractory or Relapsed Non-small Cell Lung Cancer
- PMID: 30587617
- PMCID: PMC6364089
- DOI: 10.21873/invivo.11453
Phase II Study of Weekly Amrubicin for Refractory or Relapsed Non-small Cell Lung Cancer
Abstract
Background: Amrubicin is usually administered on days 1-3 every 3 weeks by intravenous infusion. However, it causes severe hematological toxicity, especially febrile neutropenia. It was reported that weekly administration confers higher dose intensity, less severe adverse events, and anti-tumor activity that is as effective as that of treatment with a conventional schedule.
Patients and methods: Weekly amrubicin was administered at a dose of 60 mg/m2 on days 1 and 8 every 3 weeks. The primary endpoint was overall response rate.
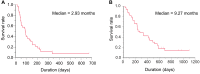
Results: A total of 33 patients were enrolled. The overall response rate was 6.1% (95% confidence interval(CI)=0.7-20.2%) and the disease control rate after 2 months was 51.5%. The median progression-free survival was 2.93 months. Febrile neutropenia was observed in only two patients.
Conclusion: The primary endpoint was not met in this study. However, weekly amrubicin achieved a high disease control rate and good tolerability.
Keywords: Amrubicin; non-small cell lung cancer; phase II; refractory; relapsed.
Copyright© 2019, International Institute of Anticancer Research (Dr. George J. Delinasios), All rights reserved.
Figures
Similar articles
-
Phase II Study of Weekly Amrubicin for Refractory or Relapsed Small Cell Lung Cancer.In Vivo. 2018 Nov-Dec;32(6):1581-1586. doi: 10.21873/invivo.11417. In Vivo. 2018. PMID: 30348719 Free PMC article. Clinical Trial.
-
Phase II study of irinotecan and amrubicin in patients with relapsed non-small cell lung cancer: Okayama Lung Cancer Study Group Trial 0402.Acta Oncol. 2012 Jul;51(6):768-73. doi: 10.3109/0284186X.2011.648342. Epub 2012 Jan 27. Acta Oncol. 2012. PMID: 22283472 Clinical Trial.
-
Phase II study of amrubicin at a dose of 45 mg/m2 in patients with previously treated small-cell lung cancer.Jpn J Clin Oncol. 2015 Oct;45(10):941-6. doi: 10.1093/jjco/hyv107. Epub 2015 Jul 31. Jpn J Clin Oncol. 2015. PMID: 26232449 Clinical Trial.
-
Phase I and pharmacokinetic study of amrubicin, a synthetic 9-aminoanthracycline, in patients with refractory or relapsed lung cancer.Cancer Chemother Pharmacol. 2006 Feb;57(3):282-8. doi: 10.1007/s00280-005-0051-2. Epub 2005 Jul 19. Cancer Chemother Pharmacol. 2006. PMID: 16028099 Clinical Trial.
-
Amrubicin for non-small-cell lung cancer and small-cell lung cancer.Invest New Drugs. 2007 Oct;25(5):499-504. doi: 10.1007/s10637-007-9069-0. Epub 2007 Jul 13. Invest New Drugs. 2007. PMID: 17628745 Review.
Cited by
-
Efficacy of weekly amrubicin for refractory or relapsed non-small cell lung cancer: A protocol of systematic review and meta-analysis.Medicine (Baltimore). 2020 Jun 19;99(25):e20454. doi: 10.1097/MD.0000000000020454. Medicine (Baltimore). 2020. PMID: 32569168 Free PMC article.
References
-
- Sugiura T, Ariyoshi Y, Negoro S, Nakamura S, Ikegami H, Takada M, Yana T, Fukuoka M. Phase I/II study of amrubicin, a novel 9-aminoanthracycline, in patients with advanced non-small-cell lung cancer. Invest New Drugs. 2005;23:331–337. - PubMed
-
- Hanada M, Noguchi T, Murayama T. Profile of the anti-tumor effects of amrubicin, a completely synthetic anthracycline. Nihon Yakurigaku Zasshi. 2003;122:141–150. - PubMed
-
- Yoshioka H, Katakami N, Okamoto H, Iwamoto Y, Seto T, Takahashi T, Sunaga N7, Kudoh S, Chikamori K, Harada M, Tanaka H, Saito H, Saka H, Takeda K, Nogami N, Masuda N, Harada T, Kitagawa H, Horio H, Yamanaka T, Fukuoka M, Yamamoto N, Nakagawa K. A randomized, open-label, phase III trial comparing amrubicin versus docetaxel in patients with previously treated non-small-cell lung cancer. Ann Oncol. 2017;28:285–291. - PubMed
-
- Inoue K, Ogawa M, Horikoshi N, Mukaiyama T, Itoh Y, Imajoh K, Ozeki H, Nagamine D, Shinagawa K. Phase I and pharmacokinetic study of SM-5887, a new anthracycline derivative. Invest New Drugs. 1989;7:213–218. - PubMed
-
- Belani CP, Ramalingam S, Perry MC, LaRocca RV, Rinaldi D, Gable PS, Tester WJ. Randomized, phase III study of weekly paclitaxel in combination with carboplatin versus standard every-3-weeks administration of carboplatin and paclitaxel for patients with previously untreated advanced non-small-cell lung cancer. J Clin Oncol. 2008;26:468–473. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous